-
Posts
8,073 -
Joined
-
Last visited
-
Days Won
551
Content Type
Profiles
Forums
Events
Blogs
Gallery
Articles
Media Demo
Store
Posts posted by MaryO
-
-
In 2021, I had the bestest ever day...

and, surpassing that:

-
 1
1
-
-
Abstract
Cushing's syndrome (CS) arises from an excess of endogenous or exogenous cortisol, with Cushing's disease specifically implicating a pituitary adenoma and exaggerated adrenocorticotropic hormone (ACTH) production. Typically, Cushing's disease presents with characteristic symptoms such as weight gain, central obesity, moon face, and buffalo hump.
This case report presents an unusual manifestation of CS in a 48-year-old male with a history of hypertension, where severe hypokalemia was the primary presentation. Initial complaints included bilateral leg swelling, muscle weakness, occasional shortness of breath, and a general feeling of not feeling well. Subsequent investigations revealed hypokalemia, metabolic alkalosis, and an abnormal response to dexamethasone suppression, raising concerns about hypercortisolism. Further tests, including 24-hour urinary free cortisol and ACTH testing, confirmed significant elevations. Brain magnetic resonance imaging (MRI) identified a pituitary macroadenoma, necessitating neurosurgical intervention.
This case underscores the rarity of CS presenting with severe hypokalemia, highlighting the diagnostic challenges and the crucial role of a collaborative approach in managing such intricate cases.
Introduction
Cushing's syndrome (CS), characterized by excessive cortisol production, is well-known for its diverse and often conspicuous clinical manifestations. Cushing's disease is a subset of CS resulting from a pituitary adenoma overproducing adrenocorticotropic hormone (ACTH), leading to heightened cortisol secretion. The classic presentation involves a spectrum of symptoms such as weight gain, central obesity, muscle weakness, and mood alterations [1].
Despite its classic presentation, CS can demonstrate diverse and atypical features, challenging conventional diagnostic paradigms. This case report sheds light on a rare manifestation of CS, where severe hypokalemia was the primary clinical indicator. Notably, instances of CS prominently manifesting through severe hypokalemia are scarce in the literature [1,2].
Through this exploration, we aim to provide valuable insights into the diagnostic intricacies of atypical CS presentations, underscore the significance of a comprehensive workup, and emphasize the collaborative approach essential for managing such uncommon hormonal disorders.
Case Presentation
A 48-year-old male with a history of hypertension presented to his primary care physician with complaints of bilateral leg swelling, occasional shortness of breath, dizziness, and a general feeling of malaise persisting for 10 days. The patient reported increased water intake and urinary frequency without dysuria. The patient was diagnosed with hypertension eight months ago. He experienced progressive muscle weakness over two months, hindering his ability to perform daily activities, including using the bathroom. The primary care physician initiated a blood workup that revealed severe hypokalemia with a potassium level of 1.3 mmol/L (reference range: 3.6 to 5.2 mmol/L), prompting referral to the hospital.
Upon admission, the patient was hypertensive with a blood pressure of 180/103 mmHg, a heart rate of 71 beats/minute, a respiratory rate of 18 breaths/minute, and an oxygen saturation of 96% on room air. Physical examination revealed fine tremors, bilateral 2+ pitting edema in the lower extremities up to mid-shin, abdominal distension with normal bowel sounds, and bilateral reduced air entry in the bases of the lungs on auscultation. The blood work showed the following findings (Table 1).
Parameter Result Reference Range Potassium (K) 1.8 mmol/L 3.5-5.0 mmol/L Sodium (Na) 144 mmol/L 135-145 mmol/L Magnesium (Mg) 1.3 mg/dL 1.7-2.2 mg/dL Hemoglobin (Hb) 15.5 g/dL 13.8-17.2 g/dL White blood cell count (WBC) 13,000 x 103/µL 4.5 to 11.0 × 109/L Platelets 131,000 x 109/L 150-450 x 109/L pH 7.57 7.35-7.45 Bicarbonate (HCO3) 46 mmol/L 22-26 mmol/L Lactic acid 4.2 mmol/L 0.5-2.0 mmol/L Table 1: Blood work findings
In order to correct the electrolyte imbalances, the patient received intravenous (IV) magnesium and potassium replacement and was later transitioned to oral. The patient was also started on normal saline at 100 cc per hour. To further investigate the complaint of shortness of breath, the patient underwent a chest X-ray, which revealed bilateral multilobar pneumonia (Figure 1). He was subsequently treated with ceftriaxone (1 g IV daily) and clarithromycin (500 mg twice daily) for seven days.
With persistent abdominal pain and lactic acidosis, a computed tomography (CT) scan abdomen and pelvis with contrast was conducted, revealing a psoas muscle hematoma. Subsequent magnetic resonance imaging (MRI) depicted an 8x8 cm hematoma involving the left psoas and iliacus muscles. The interventional radiologist performed drainage of the hematoma involving the left psoas and iliacus muscles (Figure 2).
Figure 2: Magnetic resonance imaging (MRI) depicting an 8x8 cm hematoma (arrow) involving the left psoas and iliacus muscles
In light of the concurrent presence of hypokalemia, hypertension, and metabolic alkalosis, there arose concerns about Conn's syndrome, prompting consultation with endocrinology. Their recommended workup for Conn's syndrome included assessments of the aldosterone-renin ratio and random cortisol levels. The results unveiled an aldosterone level below 60 pmol/L (reference range: 190 to 830 pmol/L in SI units) and a plasma renin level of 0.2 pmol/L (reference range: 0.7 to 3.3 mcg/L/hr in SI units). Notably, the aldosterone-renin ratio was low, conclusively ruling out Conn's syndrome. The random cortisol level was notably elevated at 1334 nmol/L (reference range: 140 to 690 nmol/L).
Furthermore, a low-dose dexamethasone suppression test was undertaken due to the high cortisol levels. Following the administration of 1 mg of dexamethasone at 10 p.m., cortisol levels were measured at 9 p.m., 3 a.m., and 9 a.m. the following day. The results unveiled a persistently elevated cortisol level surpassing 1655 nmol/L, signaling an abnormal response to dexamethasone suppression and raising concerns about a hypercortisolism disorder, such as CS.
In the intricate progression of this case, the investigation delved deeper with a 24-hour urinary free cortisol level, revealing a significant elevation at 521 mcg/day (reference range: 10 to 55 mcg/day). Subsequent testing of ACTH portrayed a markedly elevated level of 445 ng/L, distinctly exceeding the normal reference range of 7.2 to 63.3 ng/L. A high-dose 8 mg dexamethasone test was performed to ascertain the source of excess ACTH production. The baseline serum cortisol levels before the high-dose dexamethasone suppression test were 1404 nmol/L, which decreased to 612 nmol/L afterward, strongly suggesting the source of excess ACTH production to be in the pituitary gland.
A CT scan of the adrenal glands ruled out adrenal mass, while an MRI of the brain uncovered a 1.3x1.3x3.2 cm pituitary macroadenoma (Figure 3), leading to compression of adjacent structures. Neurosurgery was consulted, and they recommended surgical removal of the macroadenoma due to the tumor size and potential complications. The patient was referred to a tertiary care hospital for pituitary adenoma removal.
Discussion
CS represents a complex endocrine disorder characterized by excessive cortisol production. While the classic presentation of CS includes weight gain, central obesity, and muscle weakness, our case highlights an uncommon initial manifestation: severe hypokalemia. This atypical presentation underscores the diverse clinical spectrum of CS and the challenges it poses in diagnosis and management [1,2].
While CS typically presents with the classic symptoms mentioned above, severe hypokalemia as the initial manifestation is exceedingly rare. Hypokalemia in CS often results from excess cortisol-mediated activation of mineralocorticoid receptors, leading to increased urinary potassium excretion and renal potassium wasting. Additionally, metabolic alkalosis secondary to cortisol excess further exacerbates hypokalemia [3,4].
Diagnosing a case of Cushing's disease typically commences with a thorough examination of the patient's medical history and a comprehensive physical assessment aimed at identifying characteristic manifestations such as central obesity, facial rounding, proximal muscle weakness, and increased susceptibility to bruising. Essential to confirming the diagnosis are laboratory examinations, which involve measuring cortisol levels through various tests, including 24-hour urinary free cortisol testing, late-night salivary cortisol testing, and dexamethasone suppression tests. Furthermore, assessing plasma ACTH levels aids in distinguishing between pituitary-dependent and non-pituitary causes of CS. Integral to the diagnostic process are imaging modalities such as MRI of the pituitary gland, which facilitate the visualization of adenomas and the determination of their size and precise location [1-4].
Treatment for Cushing's disease primarily entails surgical removal of the pituitary adenoma via transsphenoidal surgery, with the aim of excising the tumor and restoring normal pituitary function. In cases where surgical intervention is unsuitable or unsuccessful, pharmacological therapies employing medications such as cabergoline (a dopamine receptor agonist) or pasireotide (a somatostatin analogue) may be considered to suppress ACTH secretion and regulate cortisol levels. Additionally, radiation therapy, whether conventional or stereotactic radiosurgery, serves as a supplementary or alternative treatment approach to reduce tumor dimensions and mitigate ACTH production [5,6]. To assess the effectiveness of treatment, manage any problem, and assure long-term illness remission, diligent long-term follow-up and monitoring are essential. Collaborative multidisciplinary care involving specialists such as endocrinologists, neurosurgeons, and other healthcare professionals is pivotal in optimizing patient outcomes and enhancing overall quality of life [2,4].
The prognosis of CS largely depends on the underlying cause, stage of the disease, and efficacy of treatment. Early recognition and prompt intervention are essential for improving outcomes and minimizing long-term complications. Surgical resection of the adrenal or pituitary tumor can lead to remission of CS in the majority of cases. However, recurrence rates vary depending on factors such as tumor size, invasiveness, and completeness of resection [2,3]. Long-term follow-up with endocrinologists is crucial for monitoring disease recurrence, assessing hormonal function, and managing comorbidities associated with CS.
Conclusions
In conclusion, our case report highlights the rarity of severe hypokalemia as the initial presentation of CS. This unique presentation underscores the diverse clinical manifestations of CS and emphasizes the diagnostic challenges encountered in clinical practice. A multidisciplinary approach involving endocrinologists, neurosurgeons, and radiologists is essential for the timely diagnosis and management of CS. Early recognition, prompt intervention, and long-term follow-up are essential for optimizing outcomes and improving the quality of life for patients with this endocrine disorder.
References
- Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM: The diagnosis of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008, 93:1526-40. 10.1210/jc.2008-0125
- Newell-Price J, Bertagna X, Grossman AB, Nieman LK: Cushing's syndrome. Lancet. 2006, 367:1605-17. 10.1016/S0140-6736(06)68699-6
- Torpy DJ, Mullen N, Ilias I, Nieman LK: Association of hypertension and hypokalemia with Cushing's syndrome caused by ectopic ACTH secretion: a series of 58 cases. Ann N Y Acad Sci. 2002, 970:134-44. 10.1111/j.1749-6632.2002.tb04419.x
- Elias C, Oliveira D, Silva MM, Lourenço P: Cushing's syndrome behind hypokalemia and severe infection: a case report. Cureus. 2022, 14:e32486. 10.7759/cureus.32486
- Fleseriu M, Petersenn S: Medical therapy for Cushing's disease: adrenal steroidogenesis inhibitors and glucocorticoid receptor blockers. Pituitary. 2015, 18:245-52. 10.1007/s11102-014-0627-0
- Pivonello R, De Leo M, Cozzolino A, Colao A: The treatment of Cushing's disease. Endocr Rev. 2015, 36:385-486. 10.1210/er.2013-1048
-
A couple years later and "My Dream Day" is substantially the same but now it would include playing with my grandchildren and, instead of practicing the aerophone, practicing the balalaika.
Today is already not "My Dream Day" since I'm posting this at 4:26 in the morning!

Maybe next year...
-
 1
1
-
-
-
Abstract
Cushing syndrome (CS) is a rare endocrinological disorder resulting from chronic exposure to excessive cortisol. The term Cushing disease is used specifically when this is caused by excessive secretion of adrenocorticotropic hormone (ACTH) by a pituitary tumor, usually an adenoma. This disease is associated with a poor prognosis, and if left untreated, it has an estimated 5-year survival rate of 50%.
We present the case of a 66-year-old female patient who received a referral to endocrinology for an evaluation of obesity due to right knee arthropathy. Taking into consideration her age, she was screened for osteoporosis, with results that showed diminished bone density. Considering this, combined with other clinical features of the patient, suspicion turned toward hypercortisolism. Laboratory findings suggested that the CS was ACTH-dependent and originated in the pituitary gland.
After a second look at the magnetic resonance imaging results, a 4-mm lesion was identified on the pituitary gland, prompting a transsphenoidal resection of the pituitary adenoma.
Introduction
Chronic excessive exposure to glucocorticoids leads to the diverse clinical manifestations of Cushing syndrome (CS), which has an annual incidence ranging from 1.8 to 3.2 cases per million individuals [1]. The syndrome's signs and symptoms are not pathognomonic, and some of its primary manifestations, such as obesity, hypertension, and glucose metabolism alterations, are prevalent in the general population [2], making diagnosis challenging. Endogenous CS falls into 2 categories: adrenocorticotropic hormone (ACTH)-dependent (80%-85% of cases), mostly due to a pituitary adenoma, or ACTH-independent (15%-20% of cases), typically caused by adrenal adenomas or hyperplasia [3]. Cushing disease (CD) represents a specific form of CS, characterized by the presence of an ACTH-secreting pituitary tumor [1]. Untreated CD is associated with high morbidity and mortality compared to the general population [1], with a 50% survival rate at 5 years [2]. However, surgical removal of a pituitary adenoma can result in complete remission, with mortality rates similar to those of the general population [2]. This article aims to highlight the challenges of suspecting and diagnosing CD and to discuss the current management options for this rare condition.
Case Presentation
A 66-year-old woman received a referral to endocrinology for an evaluation of obesity due to right knee arthropathy. During physical examination, she exhibited a body mass index of 34.3 kg/m2, blood pressure of 180/100, a history of non-insulin-requiring type 2 diabetes mellitus with glycated hemoglobin (HbA1c) of 6.9% (nondiabetic: < 5.7%; prediabetic: 5.7% to 6.4%; diabetic: ≥ 6.5%) and hypertension. Additionally, the patient complained of proximal weakness in all 4 limbs.
Diagnostic Assessment
Upon admission, densitometry revealed osteoporosis with T scores of −2.7 in the lumbar spine and −2.8 in the femoral neck. Hypercortisolism was suspected due to concomitant arterial hypertension, central obesity, muscle weakness, and osteoporosis. Physical examination did not reveal characteristic signs of hypercortisolism, such as skin bruises, flushing, or reddish-purple striae. Late-night salivary cortisol (LNSC) screening yielded a value of 8.98 nmol/L (0.3255 mcg/dL) (reference value [RV] 0.8-2.7 nmol/L [0.029-0.101 mcg/dL]) and ACTH of 38.1 pg/mL (8.4 pmol/L) (RV 2-11 pmol/L [9-52 pg/mL]). A low-dose dexamethasone suppression test (LDDST) was performed (cutoff value 1.8 mcg/dL [49 nmol/L]), with cortisol levels of 7.98 mcg/dL (220 nmol/L) at 24 hours and 20.31 mcg/dL (560 nmol/L) at 48 hours. Subsequently, a high-dose dexamethasone suppression test (HDDST) was conducted using a dose of 2 mg every 6 hours for 2 days, for a total dose of 16 mg, revealing cortisol levels of 0.0220 nmol/L (0.08 ng/mL) at 24 hours and 0.0560 nmol/L (0.0203 ng/mL) at 48 hours, alongside 24-hour urine cortisol of 0.8745 nmol/L (0.317 ng/mL) (RV 30-145 nmol/24 hours [approximately 11-53 μg/24 hours]) [4].
These findings indicated the presence of endogenous ACTH-dependent hypercortisolism of pituitary origin. Consequently, magnetic resonance imaging (MRI) was requested, but the results showed no abnormalities. Considering ectopic ACTH production often occurs in the lung, a high-resolution chest computed tomography scan was performed, revealing no lesions.
Treatment
Upon reassessment, the MRI revealed a 4-mm adenoma, prompting the decision to proceed with transsphenoidal resection of the pituitary adenoma.
Outcome and Follow-Up
The histological analysis revealed positive staining for CAM5.2, chromogranin, synaptophysin, and ACTH, with Ki67 staining at 1%. At the 1-month follow-up assessment, ACTH levels were 3.8 pmol/L (17.2 pg/mL) and morning cortisol was 115.8621 nmol/L (4.2 mcg/dL) (RV 5-25 mcg/dL or 140-690 nmol/L). Somatomedin C was measured at 85 ng/mL (RV 70-267 ng/mL) and prolactin at 3.5 ng/mL (RV 4-25 ng/mL). At the 1-year follow-up, the patient exhibited a satisfactory postoperative recovery. However, she developed diabetes insipidus and secondary hypothyroidism. Arterial hypertension persisted. Recent laboratory results indicated a glycated hemoglobin (HbA1c) level of 5.4%. Medications at the time of follow-up included prednisolone 5 milligrams a day, desmopressin 60 to 120 micrograms every 12 hours, losartan potassium 50 milligrams every 12 hours, and levothyroxine 88 micrograms a day.
Discussion
CD is associated with high mortality, primarily attributable to cardiovascular outcomes and comorbidities such as metabolic and skeletal disorders, infections, and psychiatric disorders [1]. The low incidence of CD in the context of the high prevalence of chronic noncommunicable diseases makes early diagnosis a challenge [2]. This case is relevant for reviewing the diagnostic approach process and highlighting the impact of the availability bias, which tends to prioritize more common diagnoses over rare diseases. Despite the absence of typical symptoms, a timely diagnosis was achieved.
Once exogenous CS is ruled out, laboratory testing must focus on detecting endogenous hypercortisolism to prevent misdiagnosis and inappropriate treatment [5]. Screening methods include 24-hour urinary free cortisol (UFC) for total cortisol load, while circadian rhythm and hypothalamic-pituitary-adrenal (HPA) axis function may be evaluated using midnight serum cortisol and LNSC [5]. An early hallmark of endogenous CS is the disruption of physiological circadian cortisol patterns, characterized by a constant cortisol level throughout the day or no significant decrease [2]. Measuring LNSC has proven to be useful in identifying these patients. The LNSC performed on the patient yielded a high result.
To assess HPA axis suppressibility, tests such as the overnight and the standard 2-day LDDST [5] use dexamethasone, a potent synthetic corticosteroid with high glucocorticoid receptor affinity and prolonged action, with minimal interference with cortisol measurement [6]. In a normal HPA axis, cortisol exerts negative feedback, inhibiting the secretion of corticotropin-releasing-hormone (CRH) and ACTH. Exogenous corticosteroids suppress CRH and ACTH secretion, resulting in decreased synthesis and secretion of cortisol. In pathological hypercortisolism, the HPA axis becomes partially or entirely resistant to feedback inhibition by exogenous steroids [5, 6]. The LDDST involves the administration of 0.5 mg of dexamethasone orally every 6 hours for 2 days, with a total dose of 4 mg. A blood sample is drawn 6 hours after the last administered dose [6]. Following the LDDST, the patient did not demonstrate suppression of endogenous corticosteroid production.
After diagnosing CS, the next step in the diagnostic pathway involves categorizing it as ACTH-independent vs ACTH-dependent. ACTH-independent cases exhibit low or undetectable ACTH levels, pointing to adrenal origin. The underlying principle is that excess ACTH production in CD can be partially or completely suppressed by high doses of dexamethasone, a response not observed in ectopic tumors [6]. In this case, the patient presented with an ACTH of 38.1 pg/mL (8.4 pmol/L), indicative of ACTH-dependent CD.
Traditionally, measuring cortisol levels and conducting pituitary imaging are standard practices for diagnosis. Recent advances propose alternative diagnostic methods such as positron emission tomography (PET) scans and corticotropin-releasing factor (CRF) tests [7]. PET scans, utilizing radioactive tracers, offer a view of metabolic activity in the adrenal glands and pituitary region, aiding in the identification of abnormalities associated with CD. Unfortunately, the availability of the aforementioned tests in the country is limited.
Once ACTH-dependent hypercortisolism is confirmed, identifying the source becomes crucial. A HDDST is instrumental in distinguishing between a pituitary and an ectopic source of ACTH overproduction [2, 6]. The HDDST involves administering 8 mg of dexamethasone either overnight or as a 2-day test. In this case, the patient received 2 mg of dexamethasone orally every 6 hours for 2 days, totaling a dose of 16 mg. Simultaneously, a urine sample for UFC is collected during dexamethasone administration. The HDDST suppressed endogenous cortisol production in the patient, suggesting a pituitary origin.
In ACTH-dependent hypercortisolism, CD is the predominant cause, followed by ectopic ACTH syndrome and, less frequently, an ectopic CRH-secreting tumor [3, 5]. With the pretest probability for pituitary origin exceeding 80%, the next diagnostic step is typically an MRI of the pituitary region. However, the visualization of microadenomas on MRI ranges from 50% to 70%, requiring further testing if results are negative or inconclusive [5]. Initial testing of our patient revealed no pituitary lesions. Following a pituitary location, ACTH-secreting tumors may be found in the lungs. Thus, a high-resolution chest computed tomography scan was performed, which yielded negative findings. Healthcare professionals must keep these detection rates in mind. In instances of high clinical suspicion, repeating or reassessing tests and imaging may be warranted [3], as in our case, ultimately leading to the discovery of a 4-mm pituitary adenoma.
It is fundamental to mention that the Endocrine Society Clinical Practice Guideline on Treatment of CS recommends that, when possible, all patients presenting with ACTH-dependent CS and lacking an evident causal neoplasm should be directed to an experienced center capable of conducting inferior petrosal sinus sampling to differentiate between pituitary and nonpituitary or ectopic cause [8]. However, in this instance, such a referral was regrettably hindered by logistical constraints.
Regarding patient outcomes and monitoring in CD, there is no consensus on defining remission criteria following tumor resection. Prolonged hypercortisolism results in suppression of corticotropes, resulting in low levels of ACTH and cortisol after surgical intervention. Typically, remission is identified by morning serum cortisol values below 5 µg/dL (138 nmol/L) or UFC levels between 28 and 56 nmol/d (10-20 µg/d) within 7 days after surgical intervention. In our case, the patient's morning serum cortisol was 115.8621 nmol/L (4.2 µg/dL), indicating remission. Remission rates in adults are reported at 73% to 76% in selectively resected microadenomas and at 43% in macroadenomas [8], highlighting the need for regular follow-up visits to detect recurrence.
Following the surgery, the patient experienced diabetes insipidus, a relatively common postoperative occurrence, albeit usually transient [8]. It is recommended to monitor serum sodium levels during the first 5 to 14 days postsurgery for early detection and management. Additionally, pituitary deficiencies may manifest following surgery. In this patient, prolactin levels were compromised, potentially impacting sexual response. However, postoperative somatomedin levels were normal, and gonadotropins were not measured due to the patient's age group, as no additional clinical decisions were anticipated based on those results. Secondary hypothyroidism was diagnosed postoperatively.
Moving forward, it is important to emphasize certain clinical signs and symptoms for diagnosing CD. The combination of low bone mineral density (Likelihood Ratio [LR] +21.33), central obesity (LR +3.10), and arterial hypertension (LR + 2.29) [9] has a higher positive LR than some symptoms considered “characteristic,” such as reddish-purple striae, plethora, proximal muscle weakness, and unexplained bruising [2, 10]. It is essential to give relevance to the signs the patient may present, emphasizing signs that have been proven to have an increased odds ratio (OR) such as osteoporosis (OR 3.8), myopathies (OR 6.0), metabolic syndrome (OR 2.7) and adrenal adenoma (OR 2.4) [9‐11]. The simultaneous development and worsening of these conditions should raise suspicion for underlying issues. Understanding the evolving nature of CD signs highlights the importance of vigilance during medical examinations, prioritizing the diagnostic focus, and enabling prompt initiation of treatment.
Recognizing the overlap of certain clinical features in CS is fundamental to achieving a timely diagnosis.
Learning Points
-
CS diagnosis is challenging due to the absence of pathognomonic signs and symptoms and the overlap of features present in many pathologies, such as metabolic syndrome.
-
Early detection of CS is crucial, given its association with high morbidity and mortality resulting from chronic exposure to glucocorticoids.
-
Recognizing the combination of low bone mineral density, obesity, hypertension, and diabetes as valuable clinical indicators is key in identifying CS.
-
Interdisciplinary collaboration is essential to achieve a comprehensive diagnostic approach.
Acknowledgments
We extend our gratitude to Pontificia Universidad Javeriana in Bogotá for providing essential resources and facilities that contributed to the successful completion of this case report. Special acknowledgment is reserved for the anonymous reviewers, whose insightful feedback significantly enhanced the quality of this manuscript during the peer-review process. Their contributions are sincerely appreciated.
Contributors
All authors made individual contributions to authorship. A.B.O. was involved in the diagnosis and management of this patient. M.A.G., J.M.H., and A.B.O. were involved in manuscript drafting and editing. All authors reviewed and approved the final draft.
Funding
This research received no public or commercial funding.
Disclosures
The authors declare that they have no conflicts of interest related to the current study.
Informed Patient Consent for Publication
Signed informed consent could not be obtained from the patient or a proxy but has been approved by the treating institution.
Data Availability Statement
Restrictions apply to the availability of some or all data generated or analyzed during this study to preserve patient confidentiality or because they were used under license. The corresponding author will on request detail the restrictions and any conditions under which access to some data may be provided.
References
1HakamiOA,AhmedS,KaravitakiN.Epidemiology and mortality of Cushing's syndrome.Best Pract Res Clin Endocrinol Metab.2021;35(1😞101521.2NiemanLK,BillerBMK,FindlingJW, et al.The diagnosis of Cushing's syndrome: an endocrine society clinical practice guideline.J Clin Endocrinol Metab.2008;93(5😞1526‐1540.3Gutiérrez RestrepoJ,Latorre SierraG,Campuzano MayaG.Síndrome de cushing.Med Lab.2009;15:411‐430.4PetersennS,Newell-PriceJ,FindlingJW, et al.High variability in baseline urinary free cortisol values in patients with Cushing's disease.Clin Endocrinol (Oxf).2014;80(2😞261‐269.5LilaAR,SarathiV,JagtapVS,BandgarT,MenonP,ShahNS.Cushing's syndrome: stepwise approach to diagnosis.Indian J Endocrinol Metab.2011;15(Suppl4😞S317‐S321.6DograP,VijayashankarNP.Dexamethasone suppression test. In: StatPearls StatPearls Publishing; 2024. Accessed January 29, 2024. http://www.ncbi.nlm.nih.gov/books/NBK542317/7MüllerOA,DörrHG,HagenB,StallaGK,von WerderK.Corticotropin releasing factor (CRF)-stimulation test in normal controls and patients with disturbances of the hypothalamo-pituitary-adrenal axis.Klin Wochenschr.1982;60(24😞1485‐1491.8NiemanLK,BillerBMK,FindlingJW, et al.Treatment of Cushing's syndrome: an endocrine society clinical practice guideline.J Clin Endocrinol Metab.2015;100(8😞2807‐2831.9AronDC.Cushing's syndrome: why is diagnosis so difficult?Rev Endocr Metab Disord.2010;11(2😞105‐116.10BraunLT,VogelF,ZoppS, et al.Whom should we screen for cushing syndrome? the Endocrine Society practice guideline recommendations 2008 revisited.J Clin Endocrinol Metab.2022;107(9😞e3723‐e3730.11SchneiderHJ,DimopoulouC,StallaGK,ReinckeM,SchopohlJ.Discriminatory value of signs and symptoms in Cushing's syndrome revisited: what has changed in 30 years?Clin Endocrinol (Oxf).2013;78(1😞153‐154.Abbreviations
-
ACTH
adrenocorticotropic hormone
-
CD
Cushing disease
-
CRH
corticotropin-releasing hormone
-
CS
Cushing syndrome
-
HDDST
high-dose dexamethasone suppression test
-
HPA
hypothalamic-pituitary-adrenal
-
LDDST
low-dose dexamethasone suppression test
-
LNSC
late-night salivary cortisol
-
MRI
magnetic resonance imaging
-
OR
odds ratio
-
RV
reference value
-
UFC
urinary free cortisol
© The Author(s) 2024. Published by Oxford University Press on behalf of the Endocrine Society.This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted reuse, distribution, and reproduction in any medium, provided the original work is properly cited.-
 1
1
-
-
Another picture that weirdly turned into a link.
Here's the pink jeep:
http://cushieblog.files.wordpress.com/2012/04/pink-jeep.jpg?w=300&h=225
-
 1
1
-
-
Still, and always missing you, my friend.

-
The Super Sue image

-
Abstract
Background: Cushing’s disease (CD) is associated with a specific form of metabolic syndrome that includes visceral obesity, which may affect cardiovascular hemodynamics by stimulating hypercortisolism-related metabolic activity. The purpose of this study was to evaluate the relationship between obesity and the hemodynamic profile of patients with CD.Methods: This prospective clinical study involved a hemodynamic status assessment of 54 patients newly diagnosed with CD with no significant comorbidities (mean age of 41 years). The assessments included impedance cardiography (ICG) to assess such parameters as stroke index (SI), cardiac index (CI), velocity index (VI), acceleration index (ACI), Heather index (HI), systemic vascular resistance index (SVRI), and total arterial compliance index (TACI) as well as applanation tonometry to assess such parameters as central pulse pressure (CPP) and augmentation index (AI). These assessments were complemented by echocardiography to assess cardiac structure and function.Results: Compared with CD patients without obesity, individuals with CD and obesity (defined as a body mass index ≥ 30 kg/m2) exhibited significantly lower values of ICG parameters characterizing the pumping function of the heart (VI: 37.0 ± 9.5 vs. 47.2 ± 14.3 × 1*1000−1*s−1, p = 0.006; ACI: 58.7 ± 23.5 vs. 76.0 ± 23.5 × 1/100/s2, p = 0.005; HI: 11.1 ± 3.5 vs. 14.6 ± 5.5 × Ohm/s2, p = 0.01), whereas echocardiography in obese patients showed larger heart chamber sizes and a higher left ventricular mass index. No significant intergroup differences in blood pressure, heart rate, LVEF, GLS, TACI, CPP, or AI were noted.Conclusions: Hemodynamic changes associated with obesity already occur at an early stage of CD and manifest via significantly lower values of the ICG parameters illustrating the heart’s function as a pump, despite the normal function of the left ventricle in echocardiography.1. Introduction
Cushing’s disease (CD), caused by a pituitary neuroendocrine tumor, leads to a specific type of metabolic syndrome that includes hypertension, obesity, impaired glucose metabolism, and dyslipidemia [1,2,3]. Chronic hypercortisolemia in patients with CD results in the excessive accumulation of visceral fat due to abnormal adipokine production [4]. Visceral obesity plays an important role in hypercortisolism-induced metabolic abnormalities and increased activity of the renin–angiotensin–aldosterone system activity in patients with CD [1,2,3,4,5]. Visceral obesity in patients with CD not only contributes to metabolic syndrome, but it is also an independent risk factor for cardiovascular disease [1,3,6,7]. Importantly, the structure and function of adipose tissue in patients with CD differ from those of healthy individuals [1,8,9]. The various hypercortisolism-induced metabolic abnormalities occurring in obese patients with CD may affect cardiovascular hemodynamics. There are no data on the effect of obesity on the hemodynamic profile of patients with CD and also few data are known on the association between obesity and hemodynamic disturbances in people without CD [10,11]. It was shown that the hemodynamic profile of a person with obesity is characterized by increased cardiac output and thoracic fluid content and decreased vascular resistance in comparison with these parameters in healthy individuals [12].More studies are needed to enhance our understanding of the pathophysiology of CD-related obesity as a modifiable cardiovascular risk factor, in order to develop effective preventive and therapeutic strategies. Unfortunately, subclinical consequences of hypercortisolism in newly diagnosed patients with early CD, particularly with comorbid obesity, may be undetectable with standard methods. Therefore, novel and easy-to-use diagnostic methods would be of additive value to the standard methods of assessing cardiovascular structure and function in patients with CD. A detailed evaluation of the nature of obesity in patients with CD by innovative noninvasive diagnostic methods, such as impedance cardiography (ICG), applanation tonometry (AT), and echocardiographic assessment of global longitudinal strain (GLS), may provide additional data on cardiovascular hemodynamics, particularly the heart’s pumping function, preload, and afterload [13,14,15,16,17,18]. Our previous studies demonstrated the usefulness of ICG in identifying subclinical cardiovascular complications in patients with CD [19,20].The purpose of this analysis was to assess the relationship between obesity and the hemodynamic profile of patients newly diagnosed with CD with no significant comorbidities.2. Materials and Methods
2.1. Study Population
This was a prospective observational cohort study involving a comprehensive assessment of 54 patients (mean age of 41 years) newly diagnosed with CD with no significant comorbidities (although 64.8% were diagnosed with hypertension). These patients were admitted to the Military Institute of Medicine—National Research Institute between 2016 and 2021 in order to undergo a thorough cardiovascular assessment prior to transsphenoidal pituitary neuroendocrine tumor resection surgery.This study was approved by the ethics committee at the Military Institute of Medicine—National Research Institute (approval No. 76/WIM/2016) and compliant with the Declaration of Helsinki and Good Clinical Practice guidelines. Each patient received detailed information on the purpose of this study and signed an informed consent form. This study was financed by the Polish Ministry of Research and Higher Education/Military Institute of Medicine—National Research Institute in Warsaw (grant No. 453/WIM).2.2. Inclusion Criteria
The diagnosis of CD was established based on the presence of the typical (clinical and hormonal) evidence of hypercortisolism with no adrenocorticotropic hormone (ACTH) response to corticotropin-releasing hormone (CRH) stimulation, which meets the current guidelines for the diagnosis and treatment of CD [21,22,23]. Physical examination findings consistent with the signs and symptoms of CD, including central obesity with the characteristic altered body fat distribution (a moon face and a short, thick neck); muscle atrophy in the torso and limbs; purplish stretch marks on the abdomen, hips, and thighs; thinned skin; ecchymoses; signs and symptoms of hyperandrogenism; bone pain; frequent infections; erectile dysfunction in men; and secondary amenorrhea and infertility in women. Hormone test results included elevated 24 h urinary free cortisol levels, increased morning serum cortisol levels, altered circadian rhythmicity of ACTH and cortisol secretion, elevated or detectable morning serum ACTH, and a lack of overnight serum cortisol suppression to <1.8 mg/dL during a low-dose dexamethasone suppression test (1 mg or 2 mg of dexamethasone administered at midnight). In order to ensure a pituitary etiology of CD, all patients underwent a two-day high-dose (2 mg every 6 h = a total of 8 mg) dexamethasone suppression test (HDDST), which was expected to show low serum cortisol or a >50% decrease in urinary-free cortisol levels. Moreover, each patient was shown to have no ACTH secretion response to a CRH stimulation test (with 100 μg intravenous CRH), and the presence of a pituitary neuroendocrine tumor was confirmed via contrast magnetic resonance imaging of the pituitary. Patients with inconclusive hormone tests or imaging studies additionally underwent bilateral inferior petrosal sinus sampling (used to determine ACTH levels in the venous blood before and after CRH stimulation) [21,22,23].2.3. Exclusion Criteria
The following comorbidities, which might considerably affect hemodynamic profiles, constituted our study exclusion criteria: (1) heart failure with mildly reduced or reduced left ventricular ejection fraction (LVEF) (i.e., LVEF of <50%); (2) cardiomyopathy; (3) clinically significant valvular heart disease or arrhythmia; (4) coronary artery disease, including a history of acute coronary syndrome; (5) a poor acoustic window on echocardiography; (6) a history of pulmonary embolism; (7) a history of a stroke or transient ischemic attack; (8) renal failure (estimated glomerular filtration rate < 60 mL/min/1.73 m2); (9) peripheral vascular disease and polyneuropathy; (10) chronic obstructive pulmonary disease; (11) respiratory failure (decreased partial pressure of arterial oxygen [PaO2] < 60 mmHg and/or increased partial pressure of carbon dioxide [PaCO2] > 45 mmHg); (12) a history of head trauma; (13) pregnancy; (14) age < 18 years; (15) no written informed consent.2.4. Additional Hormone Tests
Due to the fact that hypercortisolemia inhibits gonadotropin release, hormone testing was expanded to include follicle-stimulating hormone and luteinizing hormone levels. The patients also had their serum thyroid-stimulating hormone levels tested to determine possible hypothyroidism, associated with reduced CRH and thyroid-stimulating hormone secretion and hypercortisolism-induced alterations in thyroid function. The patients with CD included in this study were not receiving any medications affecting the hypothalamus–pituitary–adrenal axis. None of the female patients with CD were pregnant at the time of the study or had given birth within the previous five years.2.5. Laboratory Tests
In order to detect possible metabolic conditions, such as impaired fasting glucose, type 2 diabetes mellitus, or dyslipidemia, all patients underwent fasting blood tests from venous blood samples collected in the morning (at 6:00 a.m.). The tests evaluated the levels of fasting glucose, creatinine, eGFR, total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and triglycerides, as well as a complete blood count.2.6. Anamnesis and Physical Examination
The patients were thoroughly evaluated for cardiovascular risk factors, cardiovascular signs and symptoms, a family history of cardiovascular disease, comorbidities, prescription medications and other drugs, and smoking.The body mass index (BMI) was calculated, and obesity was determined based on the International Diabetes Federation and European Society of Cardiology guidelines, which define it as a BMI of ≥30 kg/m2 [24,25]. In the study, patients were divided into two groups: patients with CD and obesity (defined as high body mass index ≥ 30 kg/m2) and patients with CD without obesity (defined as normal BMI < 30 kg/m2).Physical examination included the resting heart rate (HR), systolic and diastolic blood pressure, and anthropometric parameters.Office blood pressure measurements were taken by a trained nurse in seated patients in the morning, after a 5 min rest. The blood pressure monitor used was Omron M4 Plus (Omron Healthcare Co. Ltd., Kyoto, Japan), which meets the European Society of Cardiology criteria [26].2.7. Echocardiography
Two-dimensional echocardiography included standard parasternal, apical, and subcostal views with a 2.5 MHz transducer (VIVID E95, GE Medical System, Wauwatosa, WI, USA) in accordance with the American Society of Echocardiography (ASE) and the European Association of Cardiovascular Imaging (EACVI) guidelines [27]. The parasternal long-axis view was used to measure the left ventricular end-diastolic diameter (LVEDd), right ventricular end-diastolic diameter (RVEDd), interventricular septal thickness, and left atrial (LA) diameter. Linear 2-dimensional left ventricular measurements were used to calculate the left ventricular mass index (LVMI), which is the left ventricular mass divided by the body surface area (LVMI cut-off values of >115 g/m2 for men and >95 g/m2 for women meet ASE and EACVI criteria for the diagnosis of left ventricular hypertrophy). The LVEF was calculated with the biplane Simpson method, based on 2-dimensional views of the left ventricle during systole and diastole in four- and two-chamber apical views. The ascending aortic diameter, valvular structure and function, and pericardium were assessed. The patients were assessed for left ventricular diastolic dysfunction according to current guidelines. Pulse wave Doppler in an apical four-chamber view aligned with mitral valve tips was used to visualize mitral inflow, including the early passive blood inflow (E) and the later atrial (A) contribution to the mitral inflow, E/A ratio, and early mitral inflow deceleration time. Apical four-chamber views were used to determine the septal and lateral early diastolic mitral annular velocities (e′ avg), and the E/e′ avg ratio was calculated [27,28].Global longitudinal strain (GLS) was assessed via electrocardiography-gated automated function imaging in two-, three-, and four-chamber views. The rates of >60 frames per second were used for optimal speckle-tracking strain assessment. Patients with a poor acoustic window were excluded from the study. Semiautomated endocardial border detection was initiated by manually selecting two points identifying the mitral annulus and one point at the apex. Segmental and whole-chamber strain was assessed. The results have been presented in the form of a “bull’s eye” graph. The data were analyzed for four-, three-, and two-chamber views, and average GLS was calculated [29].2.8. Impedance Cardiography
Based on the phenomenon of impedance variability in individual body segments associated with regional arterial blood flow, ICG is a noninvasive tool for assessing cardiovascular hemodynamics. ICG assessments were conducted by a trained nurse with a Niccomo device (Medis, Ilmenau, Germany) in patients who had been resting for 10 min in a supine position. ICG data were recorded during a 10 min assessment and processed with dedicated software (Niccomo Software, Medis). We analyzed the mean values of the following hemodynamic parameters reflecting the pumping function of the heart: (1) stroke volume (SV [mL]) and stroke index (SI [mL/m2]), based on the following formula: SV = VEPT × (dZmax/Z0) × LVET, where VEPT is tissue volume calculated from body weight, height, and patient sex, Z0 is the initial thoracic impedance, dZmax is the maximum change in thoracic impedance, and LVET is the left ventricular ejection time; (2) cardiac output (CO [mL] = SV × HR), and cardiac index (CI [mL*m−2*min−1]); (3) velocity index (VI [1*1000−1*s−1]); (4) acceleration index (ACI [1/100/s2], which is the peak acceleration of blood flow in the aorta; and (5) Heather index (HI [Ohm/s2] = dZmax × TRC, where TRC the time interval between the R-peak in the electrocardiogram and the C-point on the impedance wave). We also conducted a detailed analysis of the following afterload parameters: (1) systemic vascular resistance (SVR [dyn*s*cm−5]) together with SVR index (SVRI [dyn*s*cm−5*m2]) and (2) total arterial compliance (TAC) and TAC index (TACI [mL/mmHg] = SV/pulse pressure [mL/mmHg*m2]). Preload was assessed based on thoracic fluid content (TFC [1/kOhm], based on the formula TFC = 1000/Z0, where Z0 is the initial thoracic impedance [30,31,32].2.9. Applanation Tonometry
Applanation tonometry is a novel method of indirectly illustrating arterial pressure waveform in the aorta and arterial stiffness, which reflect left ventricular afterload. AT parameters were assessed noninvasively with a SphygmoCor system (AtCor Medical, Sydney, NSW, Australia). The measurements were taken in supine patients by a qualified nurse immediately after ICG. Radial artery pressure curves were recorded via AT with a micromanometer (Millar Instruments, Houston, TX, USA) strapped onto the left wrist. We selected high-quality recordings for our analysis. Radial pulse was calibrated against the latest brachial systolic and diastolic blood pressure measurement with an oscillometric module of the Niccomo device. SphygmoCor software (version 9.0; AtCor Medical Inc. Pty Ltd., Sydney, NSW, Australia) was used to process the arterial waveform and generate an appropriate aortic blood pressure curve from the radial pulse curve. The analyzed waveforms were composed of the pulse wave generated by the aorta and were augmented by an overlapping reflected wave. Our analyses yielded the following parameters: central systolic blood pressure; central diastolic blood pressure; central pulse pressure (CPP); augmentation pressure, which is the absolute increase in aortic systolic pressure (directly generated by left ventricular contraction) resulting from the reflection wave; and the augmentation index, calculated as AP × 100/CPP, which is a quotient of the augmentation pressure and the blood pressure in the aorta [33].2.10. Statistical Analysis
For the statistical analysis of the results, we used MS Office Excel 2023 and Statistica 12.0 (StatSof Inc., Tulsa, OK, USA). Data distribution and normality were assessed visually on histograms and with the use of the Kolmogorov–Smirnov test. Continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range, IQR), and categorical variables were expressed as absolute and relative (percentage) values. In order to evaluate differences between the subgroups of CD patients with and without comorbid obesity, we used Student’s t-test for normally distributed data, and the Mann–Whitney U test for non-normally distributed data. A comparative analysis with the use of the Mann–Whitney U test was conducted on the data from patients stratified into two subgroups: patients with CD and obesity (BMI ≥ 30 kg/m2, n = 22) and patients with CD without obesity (BMI < 30 kg/m2, n = 32). The relationship between selected indices of cardiovascular function and obesity (represented as BMI) was analyzed separately for each one in a multivariable regression model, adjusting for age and hypertension as potential covariates related to hemodynamics. The threshold of statistical significance was adopted at p < 0.05.3. Results
3.1. Baseline Characteristics
Nearly half of the patients with CD were found to be obese (n = 22, 40.7%). Overall, 20 of the 54 patients with Cushing’s disease (37%) were diagnosed with type 2 diabetes mellitus, 5 (9.3%) had prediabetes, and 29 (46.3%) had normal glucose tolerance. Of the patients with Cushing’s disease and type 2 diabetes, 14 received metformin, 5 received metformin with insulin, and 1 received insulin.The mean age, HR, hemoglobin, creatinine, and sex distribution were similar in the subgroup with and without obesity (Table 1).Table 1. Clinical, echocardiographic, hemodynamic, and applanation tonometry variables in patients with Cushing’s disease (CD) and with or without obesity.
3.2. Echocardiographic Assessment
Patients with CD and obesity (BMI ≥ 30 kg/m2) showed larger dimensions of heart chambers and ascending aorta (RVEDd, p < 0.001; LVEDd, p = 0.028; LA diameter, p < 0.001; aortic arch, p = 0.005) and higher rates of left ventricular mass index (LVMI, p = 0.028). We observed no significant differences between the subgroups in terms of the systolic (LVEF or GLS) or diastolic function of the left ventricle (Table 1).3.3. ICG and AT Assessment
The most noticeable differences in ICG were observed for parameters of the left ventricular function as a pump. In obese individuals, VI (p = 0.006), ACI (p = 0.005), and HI (p = 0.012) were lower, whereas the systolic time ratio (STR) was higher (p = 0.038) than those in non-obese individuals, with SI and CI comparable in both subgroups. We observed no significant differences in afterload (TACI, SVRI, CPP, or augmentation index) or preload (TFC) parameters (Table 1).3.4. Correlation Analysis
Analyzing the relationships between BMI and ICG hemodynamic parameters, we observed significant correlations, independent of sex and hypertension, between BMI and CI (R = 0.46; p < 0.001), SI (R = 0.29; p = 0.043), SVRI (−0.31; 0.028), and VI (R = −0.37; p = 0.0006)—see Table 2.Table 2. Correlations between hemodynamic parameters assessed with impedance cardiography and body mass index, adjusted for sex and hypertension in multivariable regression models.
4. Discussion
The results of our study revealed a relationship between obesity and hemodynamic profile assessed via ICG in patients newly diagnosed with active CD. The use of novel diagnostic modalities demonstrated that excessive fat accumulation in young and middle-aged patients with CD, already at the early stages of the disease, is associated with some hemodynamic changes in the cardiovascular system, which—at that stage—may still be undetectable in routine assessments. These findings support the need for the early detection of subclinical heart dysfunction in patients with CD to enable early treatment and help prevent cardiovascular complications [1,34,35,36].Occurring in 25%–100% of patients with CD, visceral obesity is one of the most common components of metabolic syndrome, often being the first sign of the disease. The duration of hypercortisolism correlates with obesity development [1,7,37,38], with chronic excessive cortisol levels being responsible for the abnormal distribution of adipose tissue [39]. The mechanisms behind this phenomenon may be due to the tissue overexpression of the 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1), which affects the pattern of excessive fat distribution in the torso, face, and neck [1,6]. Visceral obesity found in patients with CD is not only a component of metabolic syndrome but is in itself associated with increased metabolic activity, which makes it an independent cardiovascular risk factor, leading to the development of cardiovascular disease [1,4,9]. The tendency to accumulate visceral fat in patients with CD is also associated with abnormal adipokine production [4,6,40,41].Our study included patients newly diagnosed with active CD with no clinically significant cardiovascular disease. Males were underrepresented in both subgroups. The proportion of patients with hypertension was 64.8%, which is comparable with that reported by other authors [38,42,43,44] and similarly distributed between subgroups. However, the patients in our study presented well-controlled hypertension (mean blood pressure was 126/83 mmHg), usually with one or two medications. Considering both sex and hypertension as potential confounders, these variables were included in regression models evaluating correlations between hemodynamics and BMI.Similar to reports by other authors, our study showed higher SV and CO values in obese patients with CD; however, the respective indexed values (SI and CI) were comparable in obese and non-obese patients [12,45]. A more detailed ICG assessment demonstrated significant impairment of the pumping function of the heart as evidenced by lower HI, VI, and ACI values, and a higher STR value. The analysis of correlations revealed the independence of age and sex interrelation between some hemodynamic indices (CI, SI, SVRI, VI) and BMI. The paradox of the positive relation of obesity with volume indices of left ventricular function (CI and SI), which is negative with the marker of both its outflow and myocardial contractility (VI) encourages further studies investigating the (patho)physiological background of this phenomenon.These findings were detected despite the lack of echocardiographic evidence of left ventricular systolic or diastolic dysfunction.Moreover, our study showed larger heart chamber diameters and significantly higher LVMI in patients with CD and obesity, which is consistent with numerous earlier reports by other authors [46,47,48]. Nonetheless, it seems that in this case, increased heart chamber size and left ventricular hypertrophy should not be considered as only secondary to an increase in body weight. Hypercortisolism in patients with CD worsens the structural and functional condition of the heart muscle and may lead to myocardial fibrosis [48]. This results in myocardial remodeling associated with concentric left ventricular hypertrophy, which may impair left ventricular hemodynamic function, subsequently leading to myocardial dysfunction and symptomatic heart failure [49,50,51]. The effective treatment of patients with CD has been shown to normalize their serum cortisol levels and ultimately stop myocardial remodeling [47]. Therefore, the ICG-evidenced impaired pumping function of the heart may result from myocardial remodeling associated with complex metabolic and neuroendocrine changes in obese patients with CD [52]. These findings are consistent with previous reports on the adverse effect of obesity on left ventricular contractility [53,54,55,56].The potential mechanisms underlying the results of our study remain to be elucidated. An interesting perspective is represented by the cross-talk between glucocorticoid (GR) and mineralocorticoid receptors (MR) and their impact on metabolic syndrome. Excessive activation of the MR in extra-renal tissues by aldosterone or glucocorticoids depending on the expression of 11beta-hydroxysteroid dehydrogenase type 2 has been shown to be associated with the development of vascular dysfunction and metabolic abnormalities, leading to obesity and metabolic syndrome. High concentrations of aldosterone may also activate the transcriptional function of the GR. These mechanisms result in an interaction between GR and MR in the regulation of adipogenesis [57].The novelty of our approach is due to the use of noninvasive tools (ICG, AT) for hemodynamic assessment of the cardiovascular system in patients with CD to detect subclinical changes associated with obesity. On the one hand, our findings support earlier observations in other patient groups; on the other hand, they cast a new light on the relationship between obesity and an impaired hemodynamic profile in CD, which may result in the early development of cardiovascular complications.4.1. Clinical Implications
We determined that a dysfunctional pumping action of the heart is the key marker of impaired cardiovascular hemodynamics in obese patients newly diagnosed with CD. The use of noninvasive diagnostic methods in this study revealed a complex relationship between obesity-related hemodynamic changes and the efficiency of left ventricular contractions. An early assessment of a patient’s hemodynamic profile may help detect subclinical cardiovascular dysfunction. Such a personalized approach may facilitate early therapeutic intervention and monitoring of treatment effectiveness focused on preventing myocardial remodeling and heart dysfunction.4.2. Limitations
One limitation of our study was the small sample size. This was a result of the relatively low incidence of pituitary neuroendocrine tumors secreting ACTH. The exclusion of patients with clinically significant comorbidities further diminished the study population. However, this helped to eliminate the effect of additional factors on hemodynamic profiles. The patients assessed in our study were mostly young and middle-aged individuals with CD; therefore, our conclusions should not be extrapolated to older subjects. Although we conducted neither cardiac stress tests nor coronary angiography to exclude asymptomatic ischemic heart disease, other thorough assessments showed no physical, electrocardiographic, or echocardiographic evidence suggesting myocardial ischemia. Another potential limitation of our study is the fact that some patients had hypertension; however, it was well controlled with medications. The hemodynamic assessments involved the use of noninvasive methods as an alternative to the more expensive and less readily available invasive techniques. Nonetheless, we acknowledge the fact that noninvasive measurements can only provide indirect measurements and depend on the patient’s condition, which may vary over time.5. Conclusions
The results of our study support the usefulness of ICG in diagnosing early heart dysfunction associated with obesity in patients with CD. Asymptomatic impairment of the heart’s pumping function seems to be the earliest clinical sign of cardiovascular hemodynamic abnormalities, which at this stage are still undetectable with standard echocardiography. Individual hemodynamic profile assessment with novel noninvasive diagnostic methods encourages further studies on cardiovascular system function in obese individuals with CD and on the use of personalized therapies, which aim at preventing adverse cardiovascular events.Author Contributions
Conceptualization, A.J. and P.K.; methodology, A.J., P.K., G.G., B.U.-Ż., P.W. and G.Z.; software, P.K.; validation, A.J., P.K., B.U.-Ż., P.W. and G.Z.; formal analysis, P.K., P.W., G.G. and G.Z.; investigation, A.J., P.K., B.U.-Ż., P.W. and G.Z.; resources, A.J., P.K., B.U.-Ż., P.W. and G.Z.; data curation, A.J., P.K., B.U.-Ż., P.W., G.Z., A.K., R.W. and M.B.; writing—original draft preparation, A.J. and P.K.; writing—review and editing, G.G., B.U.-Ż., P.W. and G.Z.; visualization, A.J.; supervision, G.G. and G.Z.; project administration, G.Z.; funding acquisition, G.Z. All authors have read and agreed to the published version of the manuscript.Funding
This research was funded by the Polish Ministry of Research and Higher Education/Military Institute of Medicine—National Research Institute in Warsaw (grant No. 453/WIM).Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines and approved by the Bioethics Committee at the Military Institute of Medicine—National Research Institute in Warsaw, Poland (approval No. 76/WIM/2016; 21 December 2016).Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.Data Availability Statement
The data presented in this study are available upon request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.Acknowledgments
We would like to thank the medical personnel of the Military Institute of Medicine—National Research Institute in Warsaw for the provided patient care.Conflicts of Interest
The authors declare no conflicts of interest.References
- Pivonello, R.; Isidori, A.M.; De Martino, M.C.; Newell-Price, J.; Biller, B.M.; Colao, A. Complications of Cushing’s syndrome: State of the art. Lancet Diabetes Endocrinol. 2016, 4, 611–629. [Google Scholar] [CrossRef]
- Dekkers, O.M.; Horváth-Puhó, E.; Jørgensen, J.O.; Cannegieter, S.C.; Ehrenstein, V.; Vandenbroucke, J.P.; Pereira, A.M.; Sørensen, H.T. Multisystem morbidity and mortality in Cushing’s syndrome: A cohort study. J. Clin. Endocrinol. Metab. 2013, 98, 2277–2284. [Google Scholar] [CrossRef] [PubMed]
- Pivonello, R.; Faggiano, A.; Lombardi, G.; Colao, A. The metabolic syndrome and cardiovascular risk in Cushing’s syndrome. Endocrinol. Metab. Clin. N. Am. 2005, 34, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Pivonello, R.; De Leo, M.; Vitale, P.; Cozzolino, A.; Simeoli, C.; De Martino, M.C.; Lombardi, G.; Colao, A. Pathophysiology of diabetes mellitus in Cushing’s syndrome. Neuroendocrinology 2010, 1, 77–81. [Google Scholar] [CrossRef]
- Davy, K.P.; Hall, J.E. Obesity and hypertension: Two epidemics or one? Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R803–R813. [Google Scholar] [CrossRef]
- Lee, M.J.; Pramyothin, P.; Karastergiou, K.; Fried, S.K. Deconstructing the roles of glucocorticoids in adipose tissue biology and the development of central obesity. Biochim. Biophys. Acta 2014, 1842, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Mancini, T.; Kola, B.; Mantero, F.; Boscaro, M.; Arnaldi, G. High cardiovascular risk in patients with Cushing’s syndrome according to 1999 WHO/ISH guidelines. Clin. Endocrinol. 2004, 61, 768–777. [Google Scholar] [CrossRef]
- Bujalska, I.J.; Kumar, S.; Stewart, P.M. Does central obesity reflect “Cushing’s disease of the omentum”? Lancet 1997, 349, 1210–1213. [Google Scholar] [CrossRef]
- Galton, D.J.; Wilson, J.P. Lipogenesis in adipose tissue of patients with obesity and Cushing’s disease. Clin. Sci. 1972, 43, 17P. [Google Scholar] [CrossRef]
- Koch, R.; Sharma, A.M. Obesity and cardiovascular hemodynamic function. Curr. Hypertens. Rep. 1999, 1, 127–130. [Google Scholar] [CrossRef]
- Raison, J. Conséquences cardiovasculaires de l’obésité associée à l’hypertension artérielle [Cardiovascular consequences of obesity associated with arterial hypertension]. Presse Med. 1992, 21, 1522–1525. [Google Scholar]
- de Simone, G.; Devereux, R.B.; Kizer, J.R.; Chinali, M.; Bella, J.N.; Oberman, A.; Kitzman, D.W.; Hopkins, P.N.; Rao, D.C.; Arnett, D.K. Body composition and fat distribution influence systemic hemodynamics in the absence of obesity: The HyperGEN Study. Am. J. Clin. Nutr. 2005, 81, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Krzesiński, P.; Gielerak, G.; Kowal, J. Kardiografia impedancyjna—Nowoczesne narzedzie terapii monitorowanej chorób układu krazenia [Impedance cardiography—A modern tool for monitoring therapy of cardiovascular diseases]. Kardiol. Pol. 2009, 67, 65–71. (In Polish) [Google Scholar]
- El-Dawlatly, A.; Mansour, E.; Al-Shaer, A.A.; Al-Dohayan, A.; Samarkandi, A.; Abdulkarim, A.; Alshehri, H.; Faden, A. Impedance cardiography: Noninvasive assessment of hemodynamics and thoracic fluid content during bariatric surgery. Obes. Surg. 2005, 15, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Eikås, J.G.; Gerdts, E.; Halland, H.; Midtbø, H.; Cramariuc, D.; Kringeland, E. Arterial Stiffness in Overweight and Obesity: Association with Sex, Age, and Blood Pressure. High Blood Press Cardiovasc. Prev. 2023, 30, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Abomandour, H.G.; Elnagar, A.M.; Aboleineen, M.W.; Shehata, I.E. Subclinical Impairment of Left Ventricular Function assessed by Speckle Tracking in Type 2 Diabetic Obese and Non-Obese Patients: Case Control Study. J. Cardiovasc. Echogr. 2022, 32, 95–106. [Google Scholar] [CrossRef]
- Galderisi, M.; Lomoriello, V.S.; Santoro, A.; Esposito, R.; Olibet, M.; Raia, R.; Di Minno, M.N.; Guerra, G.; Mele, D.; Lombardi, G. Differences of myocardial systolic deformation and correlates of diastolic function in competitive rowers and young hypertensives: A speckle-tracking echocardiography study. J. Am. Soc. Echocardiogr. 2010, 23, 1190–1198. [Google Scholar] [CrossRef]
- Kalam, K.; Otahal, P.; Marwick, T.H. Prognostic implications of global LV dysfunction: A systematic review and meta-analysis of global longitudinal strain and ejection fraction. Heart 2014, 100, 1673–1680. [Google Scholar] [CrossRef]
- Jurek, A.; Krzesiński, P.; Gielerak, G.; Witek, P.; Zieliński, G.; Kazimierczak, A.; Wierzbowski, R.; Banak, M.; Uziębło-Życzkowska, B. Cushing’s Disease: Assessment of Early Cardiovascular Hemodynamic Dysfunction With Impedance Cardiography. Front. Endocrinol. 2021, 12, 751743. [Google Scholar] [CrossRef]
- Jurek, A.; Krzesiński, P.; Uziębło-Życzkowska, B.; Witek, P.; Zieliński, G.; Kazimierczak, A.; Wierzbowski, R.; Banak, M.; Gielerak, G. The patient’s sex determines the hemodynamic profile in patients with Cushing disease. Front. Endocrinol. 2023, 14, 1270455. [Google Scholar] [CrossRef]
- Fleseriu, M.; Auchus, R.; Bancos, I.; Ben-Shlomo, A.; Bertherat, J.; Biermasz, N.R.; Boguszewski, C.L.; Bronstein, M.D.; Buchfelder, M.; Carmichael, J.D.; et al. Consensus on diagnosis and management of Cushing’s disease: A guideline update. Lancet Diabetes Endocrinol. 2021, 9, 847–875. [Google Scholar] [CrossRef] [PubMed]
- Nieman, L.K.; Biller, B.M.; Findling, J.W.; Newell-Price, J.; Savage, M.O.; Stewart, P.M.; Montori, V.M. The diagnosis of Cushing’s syndrome: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2008, 93, 1526–1540. [Google Scholar] [CrossRef]
- Ceccato, F.; Boscaro, M. Cushing’s syndrome: Screening and diagnosis. High. Blood Press. Cardiovasc. Prev. 2016, 23, 209–215. [Google Scholar] [CrossRef]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr.; et al. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Bäck, M.; Benetos, A.; Biffi, A.; Boavida, J.M.; Capodanno, D.; et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2021, 42, 3227–3337. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015, 28, 1–39.e14. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F.; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2016, 29, 277–314. [Google Scholar] [CrossRef]
- Voigt, J.U.; Pedrizzetti, G.; Lysyansky, P.; Marwick, T.H.; Houle, H.; Baumann, R.; Pedri, S.; Ito, Y.; Abe, Y.; Metz, S.; et al. Definitions for a common standard for 2D speckle tracking echocardiography: Consensus document of the EACVI/ASE/Industry Task Force to standardize deformation imaging. J. Am. Soc. Echocardiogr. 2015, 28, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, V.; Isakson, S.; Bhalla, M.A.; Lin, J.P.; Clopton, P.; Gardetto, N.; Maisel, A.S. Diagnostic Ability of B-Type Natriuretic Peptide and Impedance Cardiography: Testing to Identify Left Ventricular Dysfunction in Hypertensive Patients. Am. J. Hypertens. 2005, 18, 73S–81S. [Google Scholar] [CrossRef]
- Parrott, C.W.; Burnham, K.M.; Quale, C.; Lewis, D.L. Comparison of Changes in Ejection Fraction to Changes in Impedance Cardiography Cardiac Index and Systolic Time Ratio. Congest. Heart Fail. 2004, 10, 11–13. [Google Scholar] [CrossRef]
- Packer, M.; Abraham, W.T.; Mehra, M.R.; Yancy, C.W.; Lawless, C.E.; Mitchell, J.E.; Smart, F.W.; Bijou, R.; O’Connor, C.M.; Massie, B.M.; et al. Utility of Impedance Cardiography for the Identification of Short-Term Risk of Clinical Decompensation in Stable Patients with Chronic Heart Failure. J. Am. Coll. Cardiol. 2006, 47, 2245–2252. [Google Scholar] [CrossRef]
- Pauca, A.L.; O’Rourke, M.F.; Kon, N.D. Prospective evaluation of a method for estimating ascending aortic pressure from the radial artery pressure waveform. Hypertension 2001, 38, 932–937. [Google Scholar] [CrossRef] [PubMed]
- Coulden, A.; Hamblin, R.; Wass, J.; Karavitaki, N. Cardiovascular health and mortality in Cushing’s disease. Pituitary 2022, 25, 750–753. [Google Scholar] [CrossRef] [PubMed]
- Clayton, R.N. Cardiovascular complications of Cushings syndrome: Impact on morbidity and mortality. J. Neuroendocrinol. 2022, 34, e13175. [Google Scholar] [CrossRef] [PubMed]
- De Leo, M.; Pivonello, R.; Auriemma, R.S.; Cozzolino, A.; Vitale, P.; Simeoli, C.; De Martino, M.C.; Lombardi, G.; Colao, A. Cardiovascular disease in Cushing’s syndrome: Heart versus vasculature. Neuroendocrinology 2010, 1, 50–54. [Google Scholar] [CrossRef]
- Faggiano, A.; Pivonello, R.; Spiezia, S.; De Martino, M.C.; Filippella, M.; Di Somma, C.; Lombardi, G.; Colao, A. Cardiovascular risk factors and common carotid artery caliber and stiff ness in patients with Cushing’s disease during active disease and 1 year after disease remission. J. Clin. Endocrinol. Metab. 2003, 88, 2527–2533. [Google Scholar] [CrossRef] [PubMed]
- Witek, P.; Zieliński, G.; Szamotulska, K.; Witek, J.; Zgliczyński, W. Complications of Cushing’s disease—Prospective evaluation and clinical characteristics. Do they affect the efficacy of surgical treatment? Endokrynol. Pol. 2012, 63, 277–285. [Google Scholar] [PubMed]
- Geer, E.B.; Shen, W.; Gallagher, D.; Punyanitya, M.; Looker, H.C.; Post, K.D.; Freda, P.U. MRI assessment of lean and adipose tissue distribution in female patients with Cushing’s disease. Clin. Endocrinol. 2010, 73, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Wang, M. The role of glucocorticoid action in the pathophysiology of the metabolic syndrome. Nutr. Metab. 2005, 2, 3. [Google Scholar] [CrossRef]
- Tataranni, P.A.; Larson, D.E.; Snitker, S.; Young, J.B.; Flatt, J.P.; Ravussin, E. Eff ects of glucocorticoids on energy metabolism and food intake in humans. Am. J. Physiol. 1996, 271, E317–E325. [Google Scholar] [PubMed]
- Isidori, A.M.; Graziadio, C.; Paragliola, R.M.; Cozzolino, A.; Ambrogio, A.G.; Colao, A.; Corsello, S.M.; Pivonello, R. ABC Study Group. The hypertension of Cushing’s syndrome: Controversies in the pathophysiology and focus on cardiovascular complications. J. Hypertens. 2015, 33, 44–60. [Google Scholar] [CrossRef]
- Dekkers, O.M.; Biermasz, N.R.; Pereira, A.M.; Roelfsema, F.; van Aken, M.O.; Voormolen, J.H.; Romijn, J.A. Mortality in patients treated for Cushing’s disease is increased, compared with patients treated for nonfunctioning pituitary macroadenoma. J. Clin. Endocrinol. Metab. 2007, 92, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.K.; Goldberg, L.; Fayngold, S.; Kostadinov, J.; Post, K.D.; Geer, E.B. Predictors of mortality and long-term outcomes in treated Cushing’s disease: A study of 346 patients. J. Clin. Endocrinol. Metab. 2013, 98, 1022–1030. [Google Scholar] [CrossRef]
- Abel, E.D.; Litwin, S.E.; Sweeney, G. Cardiac remodeling in obesity. Physiol. Rev. 2008, 88, 389–419. [Google Scholar] [CrossRef]
- Hey, T.M.; Dahl, J.S.; Brix, T.H.; Søndergaard, E.V. Biventricular hypertrophy and heart failure as initial presentation of Cushing’s disease. BMJ Case Rep. 2013, bcr2013201307. [Google Scholar] [CrossRef] [PubMed]
- Toja, P.M.; Branzi, G.; Ciambellotti, F.; Radaelli, P.; De Martin, M.; Lonati, L.M.; Scacchi, M.; Parati, G.; Cavagnini, F.; Cavagnini, F. Clinical relevance of cardiac structure and function abnormalities in patients with Cushing’s syndrome before and after cure. Clin. Endocrinol. 2012, 76, 332–338. [Google Scholar] [CrossRef]
- You, K.H.; Marsan, N.A.; Delgado, V.; Biermasz, N.R.; Holman, E.R.; Smit, J.W.; Feelders, R.A.; Bax, J.J.; Pereira, A.M. Increased myocardial fibrosis and left ventricular dysfunction in Cushing’s syndrome. Eur. J. Endocrinol. 2012, 166, 27–34. [Google Scholar] [CrossRef]
- Baykan, M.; Erem, C.; Gedikli, O.; Hacihasanoglu, A.; Erdogan, T.; Kocak, M.; Kaplan, S.; Kiriş, A.; Orem, C.; Celik, S. Assessment of left ventricular diastolic function and Tei index by tissue Doppler imaging in patients with Cushing’s Syndrome. Echocardiography 2008, 25, 182–190. [Google Scholar] [CrossRef]
- Ainscough, J.F.; Drinkhill, M.J.; Sedo, A.; Turner, N.A.; Brooke, D.A.; Balmforth, A.J.; Ball, S.G. Angiotensin II type-1 receptor activation in the adult heart causes blood pressure-independent hypertrophy and cardiac dysfunction. Cardiovasc. Res. 2009, 81, 592–600. [Google Scholar] [CrossRef]
- Uziębło-Życzkowska, B.; Krzesinński, P.; Witek, P.; Zielinński, G.; Jurek, A.; Gielerak, G.; Skrobowski, A. Cushing’s Disease: Subclinical Left Ventricular Systolic and Diastolic Dysfunction Revealed by Speckle Tracking Echocardiography and Tissue Doppler Imaging. Front. Endocrinol. 2017, 8, 222. [Google Scholar] [CrossRef]
- Eschalier, R.; Rossignol, P.; Kearney-Schwartz, A.; Adamopoulos, C.; Karatzidou, K.; Fay, R.; Mandry, D.; Marie, P.Y.; Zannad, F. Features of cardiac remodeling, associated with blood pressure and fibrosis biomarkers, are frequent in subjects with abdominal obesity. Hypertension 2014, 63, 740–746. [Google Scholar] [CrossRef]
- Krzesiński, P.; Stańczyk, A.; Piotrowicz, K.; Gielerak, G.; Uziębło-Zyczkowska, B.; Skrobowski, A. Abdominal obesity and hypertension: A double burden to the heart. Hypertens. Res. 2016, 39, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.Y.; O’Moore-Sullivan, T.; Leano, R.; Byrne, N.; Beller, E.; Marwick, T.H. Alterations of left ventricular myocardial characteristics associated with obesity. Circulation 2004, 110, 3081–3087. [Google Scholar] [CrossRef] [PubMed]
- Parrinello, G.; Licata, A.; Colomba, D.; Di Chiara, T.; Argano, C.; Bologna, P.; Corrao, S.; Avellone, G.; Scaglione, R.; Licata, G. Left ventricular filling abnormalities and obesity-associated hypertension: Relationship with overproduction of circulating transforming growth factor beta1. J. Hum. Hypertens. 2005, 19, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Gao, Y.; Tan, K.; Li, P. Subclinical impairment of left ventricular function in diabetic patients with or without obesity: A study based on three-dimensional speckle tracking echocardiography. Herz 2014, 40, 260–268. [Google Scholar] [CrossRef]
- Feraco, A.; Marzolla, V.; Scuteri, A.; Armani, A.; Caprio, M. Mineralocorticoid Receptors in Metabolic Syndrome: From Physiology to Disease. Trends Endocrinol. Metab. 2020, 31, 205–217. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).-
 1
1
-

I have seen this image several places online and it never ceases to crack me up. Sometimes, we really have strange things going on inside our bodies.
Usually, unlike Kermit, we ourselves know that something isn't quite right, even before the doctors know. Keep in touch with your own body so you'll know, even before the MRI.
I asked doctors for several years - PCP, gynecologist, neurologist, podiatrist - all said the now-famous refrain. "It's too rare. You couldn't have Cushing's." I kept persisting in my reading, making copies of library texts even when I didn't understand them, keeping notes. I just knew that someone, somewhere would "discover" that I had Cushing's.
Finally, someone did.
These days, there's no excuse to keep you from learning all you can about what's going on with you. There's your computer and the internet. Keep reading and learning all you can. You have a vested interest in what's going on inside, not your doctor.

-
Boy, I really needed to reread this again today.
Many thanks again...to me!...for sharing.
Except for my new "hobby" of learning the balalaika, everything is seeming painful, blah, boring and, at times, plain annoying.
(this is my group, but was made a few years before I joined)
I have to try to remember that TODAY is The Best Day Of My Life - no matter what!

-
Abstract
In Cushing syndrome (CS), prolonged exposure to high cortisol levels results in a wide range of devastating effects causing multisystem morbidity. Despite the efficacy of treatment leading to disease remission and clinical improvement, hypercortisolism-induced complications may persist. Since glucocorticoids use the epigenetic machinery as a mechanism of action to modulate gene expression, the persistence of some comorbidities may be mediated by hypercortisolism-induced long-lasting epigenetic changes. Additionally, glucocorticoids influence microRNA expression, which is an important epigenetic regulator as it modulates gene expression without changing the DNA sequence. Evidence suggests that chronically elevated glucocorticoid levels may induce aberrant microRNA expression which may impact several cellular processes resulting in cardiometabolic disorders.
The present article reviews the evidence on epigenetic changes induced by (long-term) glucocorticoid exposure. Key aspects of some glucocorticoid-target genes and their implications in the context of CS are described. Lastly, the effects of epigenetic drugs influencing glucocorticoid effects are discussed for their ability to be potentially used as adjunctive therapy in CS.
Issue Section:Mini-reviewIn Cushing syndrome (CS), adrenocorticotropic hormone (ACTH) hypersecretion by a pituitary adenoma or an ectopic source, or autonomous cortisol hypersecretion by an adrenal tumor, induces chronic endogenous hypercortisolism with loss of the cortisol circadian rhythm (1). CS is more prevalent in women than men and frequently occurs in the fourth to sixth decades of life (2).
Glucocorticoids (GC) have extensive physiological actions and regulate up to 20% of the expressed genome, mainly related to the immune system, metabolic homeostasis, and cognition. Therefore, the prolonged exposure to high cortisol levels results in a wide range of devastating effects, including major changes in body composition (obesity, muscle atrophy, osteoporosis), neuropsychiatric disturbances (impaired cognition, depression, sleep disturbances), the metabolic syndrome (obesity, hypertension, insulin resistance, and dyslipidemia), hypercoagulability, and immune suppression (3, 4). The consequences of hypercortisolism lead to compromised quality of life and increased mortality rate (5). The mortality rate in patients with CS is 4 times higher than the healthy control population (6). Risk factors such as obesity, diabetes, and hypertension contribute to the increased risk of myocardial infarction, stroke, and cardiac insufficiency. As a result, cardiovascular disease is the leading cause of the premature death in CS (5). Infectious disease is also an important cause of death in CS (5). Therefore, prompt treatment to control hypercortisolism is imperative to prevent complications and an increased mortality rate.
Despite the efficacy of treatment leading to disease remission, the clinical burden of CS improves, but does not completely revert, in every patient (7). Indeed, obesity, neuropsychiatric disturbances, hypertension, diabetes, and osteoporosis persist in a substantial number of biochemically cured patients. For instance, in a study involving 118 CS patients in remission for about 7.8 years (median), resolution of comorbidities such as diabetes occurred in only 36% of cases, hypertension in 23% of cases, and depression in 52% of the cases (8). It has been proposed that epigenetic changes as a consequence of hypercortisolism is a mechanism of the persistence of some comorbidities (9-12).
Epigenetics is a reversible process that modifies gene expression without any alterations in DNA sequence; frequently it is mediated by histone modification and DNA methylation together with microRNAs (13-15). GCs use the epigenetic machinery as a mechanism of action to regulate gene expression in physiological circumstances, such as metabolic actions and stress response. Its networks involve DNA and histone modifying enzymes, such as DNA methyltransferases (DNMTs), histone acetyltransferases (HATs), and histone deacetylases (HDACs) (16). (Fig. 1) The DNA methylation process catalyzed by DNMTs is usually associated with downregulation of gene expression (17). Histone modifications catalyzed by HAT enzymes induce gene transcription, while those by HDAC enzymes induce transcriptional repression (17). Drugs interfering with these enzymes (so-called epigenetic drugs) may affect the GC genomic actions confirming the interaction between GC and the epigenetic system (18, 19). Furthermore, GC can modulate HDAC and DNMT expression and activity (16, 19, 20). Based on these data it might be speculated that in CS, epigenetic modifications induced by long-term GC exposure plays a role in the development of the disease-specific morbidity (9, 10).
Figure 1.Glucocorticoid (GC) and its epigenetic machinery. GC through its receptor interacts with DNA and histone modifying enzymes, such as DNA methyltransferases (DNMTs), histone acetyl transferases (HATs), and histone deacetylases (HDAC) to modulate gene expression.
In this review we provide an overview of epigenetic aspects of GC action in physiological conditions and in the context of CS. We start with a detailed characterization of how GC, using the epigenetic system, can change chromatin structure in order to activate or silence gene expression. (Fig. 2) Subsequently, we describe the role of epigenetic mechanisms in the regulation of expression of several GC-target genes related to CS. Finally, we present the current evidence of epigenetic changes caused by the long-term of GC exposure and the potential use of epidrugs influencing GC actions.
Figure 2.Epigenetic mechanisms of the glucocorticoid action to regulate gene expression. The GR is located in cytoplasm in a multi-protein complex; after GC binding, GR dissociates from the multi-protein complex, crosses the nuclear membrane, dimerizes, and binds to the GRE of the target gene. One of the mechanisms of action of GC is through the recruitment of co-regulators together with epigenetic enzymes, such as HAT, to change the chromatin structure, resulting in activation of gene transcription. Also, GR decreases gene expression by tethering other transcriptional factors and recruiting HDAC2, causing histone deacetylation, which leads to a repressed chromatin. GC can cause hypomethylation through downregulation in the expression of DNMT1. Abbreviations: Ac, acetylation; DNMT1, DNA methyltransferase 1; GC, glucocorticoid; GR, glucocorticoid receptor; GRE, glucocorticoid responsive elements; HAT, histone acetyltransferase; HDAC, histone deacetylases; Me: methylation.
Search Strategy
A search of the PubMed database was conducted using the advanced search builder tool for articles in the English language on the following terms “glucocorticoids,” “glucocorticoid receptor,” “Cushing,” “hypercortisolism,” “epigenetic,” “DNA methylation,” “histone deacetylase,” “histone acetyltransferase,” “microRNA” “fkbp5,” “clock genes,” and “POMC.” Moreover, references were identified directly from the articles included in this manuscript. The articles were selected by the authors after being carefully analyzed regarding their importance and impact.
Epigenetic Aspects of Genomic Action of Glucocorticoids
GCs regulate gene expression positively or negatively. GC-responsive genes include genes encoding for proteins associated with inflammation, metabolic processes, blood pressure and fluid homeostasis, apoptosis, cell cycle progression, circadian rhythm, and intracellular signaling (21).
The GC actions are cell type–specific (22). For instance, in an in vitro study, the comparison of GC-expressed genes between 2 cell lines, corticotroph (AtT20) and mammary (3134) cell lines, showed a different set of GC-regulated genes, revealing the cell type–specific nature of GC effects (23). GC function depends on the accessibility of glucocorticoid receptor (GR)-binding sites in the DNA of the target tissue, which in turn is mostly established during cell differentiation. Therefore, different chromatin organization explains the distinct GR-binding sites among different tissues (22, 24, 25). The chromatin accessibility is determined by histone modifications such as acetylation, methylation, phosphorylation, and/or DNA methylation, processes that are both dynamic and reversible (26).
Furthermore, gene expression is regulated in a GC-concentration-dependent manner which is tissue-specific. Only a few genes can be upregulated or downregulated at low concentrations of GC. For example, a dose of dexamethasone (Dex) as low as 0.5 nM selectively activated PER1 (period 1, transcription factor related to circadian rhythm) expression in lung cancer (A549) cells (21, 27). Additionally, continuous GC exposure or pulsed GC (cortisol fluctuation during circadian rhythm) may cause different responses with respect to gene expression (26, 28). For example, constant treatment with corticosterone induced higher levels of PER1 clock gene mRNA expression compared with pulsatile treatment, as demonstrated in an in vitro study using 3134 cell line (28).
The time course for gene expression in response to Dex is fast, with repression occurring slightly slower compared to activation. Half of activated and repressed genes are detected within, respectively, about 40 minutes and 53 minutes following Dex exposure (21).
In short, the transcriptional output in response to GC depends on cell type, as well as on the duration and intensity of GC exposure (21, 24, 26, 27). GCs act as a transcriptional regulatory factor resulting in activating or repressing the expression of genes. The GC exerts its function through binding to corticosteroid receptors, specifically, the mineralocorticoid receptor and the GR, members of the nuclear receptor superfamily (29, 30).
Glucocorticoid Receptor
The GR is located in the cytoplasm in a chaperone complex which includes heat-shock proteins (70 and 90) and immunophilins (such as FK506 binding protein [FKBP5]). Cortisol diffuses across the cell membrane and binds with high affinity to the GR. The activated GR bound to GC dissociates of the multi-protein complex and is transferred to the nucleus, where it ultimately regulates gene expression (26, 31).
GR is a transcription factor encoded by nuclear receptor subfamily 3, group C member 1 (NR3C1) gene, located in chromosome 5, and consisting of 9 exons. It is composed of 3 major functional domains, namely a DNA binding domain (DBD), the C-terminal ligand-binding domain (LBD) and the N-terminal domain (NTB). The LBD recognizes and joins the GC. NTB contains an activation function-1 (AF1) which connects with co-regulators and the members of the general transcription machinery to activate target genes. The DBD comprises 2 zinc fingers motifs that are able to identify and bind to glucocorticoid responsive elements (GREs) (32, 33).
GRα is the most expressed and functionally active GR. GRβ is another isoform which is the result of an alternative splicing in exon 9 of the GR transcript. The difference between the 2 isoforms is the distinct ligand-binding domain in GRβ. This variance prevents the GRβ from binding to GC. In fact, the GRβ counteracts GRα function by interfering with its binding to a GRE in the target gene, and GRβ expression is associated with GC resistance (32). In addition, GRβ has its own transcriptional activity which is independent and distinct from GRα (34).
Another splice variant of human GR, GRγ, is associated with GC resistance in lung cell carcinoma and childhood acute lymphoblastic leukemia (33, 35). There is an additional amino acid (arginine) in the DBD of the GRγ that reduces, by about half, the capacity to activate or suppress the transcription of the target gene, as compared with GRα (32). One study identified GRγ in a small series of corticotroph adenomas (36).
Glucocorticoid Mechanism of Action
The GR-GC complex induces or represses gene expression directly by binding to DNA, indirectly by tethering other transcription factors or yet in a composite manner that consists in binding DNA in association with binding to other co-regulators (35, 37).
The GR has the ability to reorganize the chromatin structure to become more or less accessible to the transcriptional machinery. In the classical mechanism of direct induction of gene expression, the GR dimerizes and binds to a GRE in DNA. The receptor recruits co-regulators, such as CREB binding protein, which has intrinsic histone acetyltransferase (HAT) activity that modifies the chromatin structure from an inactive to an active state. This model, called transactivation, upregulates the expression of some genes related to glucose, protein, and fat metabolism. Gene repression, on the other hand, is accomplished by GR binding to a negative GRE (nGRE) leading to the formation of a chromatin remodeling complex composed by co-repressor factors, such as NCOR1 and SMRT, and histone deacetylases (HDACs), that ultimately turn chromatin less accessible and suppress gene transcription. The gene repression through direct binding events occurs less frequently when compared to gene induction (25, 35, 38).
Another mechanism of GC action is through binding to other transcription factors (tethering). In case of switching off inflammatory genes, GR binds to transcriptional co-activator molecules, such as CREB binding protein with intrinsic HAT activity, and subsequently recruits HDAC2 to reverse histone acetylation, thus resulting in a suppression of the activated inflammatory gene (39). In the same model, GC interacts with other cofactors, such as the STAT family, to induce chromatin modifications resulting in increased gene expression (26).
Furthermore, the transcriptional dynamics of some genes follow a composite manner. In this model, GR, in conjunction with binding to GRE, also interacts with cofactors in order to enhance or reduce gene expression (35).
GCs can also modulate gene expression by influencing the transcription of epigenetic modifiers. An experimental study demonstrated that GC mediated the upregulation of HDAC2 in rats exposed to chronic stress, which in turn decreased the transcription of histone methyltransferase (Ehmt2) that ultimately upregulated the expression of Nedd4. Nedd4 is a ubiquitin ligase, expression of which has been related to cognitive impairment (40). Additionally, GC was found to interact with another epigenetic eraser, namely JMJD3, a histone demethylase, suppressing its transcription in endothelial cells treated with TNFα that led to decreased expression of other genes related to the blood-brain barrier (41).
GCs have the ability to induce (de)methylation changes in DNA, ultimately affecting gene expression. The DNA methylation process triggered by GC involves the family of DNA methyltransferases (DNMT) and ten-eleven translocation (TET) protein (20, 42-44). The DNMT, DNMT1, DNMT3A, and DNMT3B are able to transfer a methyl group to a cytosine residue in DNA, forming 5-methylcytosine (5mC), which negatively impacts gene expression. In contrast, TET protein chemically modifies the 5mC to form 5-hydroxymethylcytosine (5hmC), which ultimately leads to unmethylated cytosine, positively influencing gene expression (45).
Glucocorticoids mainly induce loss of methylation events rather than gain of methylation across the genome (11, 46). The DNA demethylation process can be either active or passive. The active mechanism is linked to the upregulation of TET enzyme expression that follows GC treatment, which was described in retinal and osteocyte cell line model studies (42, 43). The passive demethylation event involves the downregulation (Fig. 2) or dysfunction of DNMT1. DNMT1 is responsible for maintaining the methylation process in dividing cells (45). In case of GC exposure, GC can cause hypomethylation through downregulation in the expression of DNMT1, a process described in the AtT20 corticotroph tumor cell model, or through GC hindering DNMT activity, particularly DNMT1, as demonstrated in the retinal cell (RPE) line (20, 42, 44).
Glucocorticoid-Induced Epigenetic Changes
There are several molecular mechanisms connecting GR activation and epigenetic modifications ultimately affecting gene expression (Fig. 2). As described above, GC uses epigenetic machinery, such as DNA and histone modifying enzymes, to restructure the chromatin in order to induce or silence gene transcription (16, 47).
In an in vitro study using murine AtT20 corticotroph tumor and neuronal cell lines, after chronic GC exposure followed by a recovery period in the absence of GC, the cells retained an “epigenetic memory” with persistence of loss of methylation content in FKBP5 gene but with no increased gene expression at baseline. The functionality of this “epigenetic memory” only became evident in a second exposure to GC, when the cells responded sharply with a more robust expression of FKBP5 gene compared to the cells without previous exposure to GC (44). Another in vitro study, using a human fetal hippocampal cell line, confirmed long-lasting DNA methylation changes induced by GC. The cells were treated for 10 days with dexamethasone, during the proliferative and cell differentiation phases of the cell line, followed by 20 days without any treatment. The second exposure to GC resulted in an enhanced gene expression of a subset of GC-target genes (48). Additionally, using an animal model subjected to chronic stress, a distinct gene expression profile was demonstrated in response to acute GC challenge compared to those without chronic stress history. The proposed mechanism was that chronic stress resulted in GC-induced enduring epigenetic changes in target genes, altering the responsiveness to a subsequent GC exposure (49).
In general, it seems that the majority of differential methylation regions (DMRs) induced by GC are loss of methylation rather than gain of methylation. In an experimental study, an association between hypomethylation and GC exposure was demonstrated in mice previously exposed to high levels of GC. Further analysis demonstrated that the genes linked with DMR were mostly related to metabolism, the immune system, and neurodevelopment (11).
Human studies have also shown that excess of cortisol can induce modifications in DNA methylation. DNA methylation data obtained from whole blood samples from patients with chronic obstructive pulmonary disease (COPD) treated with GC revealed DMR at specific CpG dinucleotides across the genome. These DMR were confirmed by pyrosequencing and annotated to genes, such as SCNN1A, encoding the α subunit of the epithelial sodium channel, GPR97, encoding G protein coupled receptor 97, and LRP3, encoding low-density lipoprotein receptor-related protein 3 (50). Furthermore, it has been proposed that the negative impact of chronic GC exposure on the immune system, which increases the risk of opportunistically infections, may be epigenetically mediated (51). In a clinical study, using whole blood samples, an analysis of genome-wide DNA methylation was performed on patients before and after exposure to GC (51). Long-term GC exposure disrupts, through a persistent modification of the cytosine methylation pattern, the mTORC1 pathway which affects CD4+ T cell biology (51).
Taken together, these data clearly show the interplay between GC signaling and methylation and histone modifications processes suggesting that GC interferes in the epigenetic landscape modulating gene expression. It is possible that most of these GC-induced epigenetic events are dynamic and temporary, while others may persist leading to long-lasting disorders. Further research to provide insight into what makes some events reversible is warranted.
Epigenetic Changes as a Consequence of Long-Term Glucocorticoid Exposure in Cushing Syndrome
The comorbidities associated with CS are associated with increased mortality mainly due to cardiovascular events (52). GC-induced comorbidities in CS may be at least in part epigenetically mediated. Previous study using whole blood methylation profile demonstrated that specific hypomethylated CpG sites induced by GC were associated with Cushing comorbidities, such as hypertension and osteoporosis (46). The study identified a methylator predictor of GC excess which could be used as a biomarker to monitor GC status (46).
The long-term exposure to high cortisol levels may be crucial for the persistence of some morbidities in CS through epigenetic changes. Hypercortisolism-induced persistent changes in visceral adipose tissue gene expression through epigenetic modifications was investigated in a translational study (12). This study combined data from patients with active CS and data from an animal model of CS in active and remitted phase. Interestingly, the study demonstrated long-lasting changes in the transcriptome of adipose tissue that were associated with histone modifications induced by GC. Therefore, these epigenetic fingerprints observed even after the resolution of hypercortisolism may elucidate the mechanism of persistent modifications in gene expression in the visceral adipose tissue (12).
With regard to the persistence of GC-induced DMR, a genome-wide DNA methylation analysis showed a lower average of DNA methylation in patients in remission of CS compared to controls. Interestingly, the most common biologically relevant affected genes were retinoic acid receptors, thyroid hormone receptors, or hormone/nuclear receptors, important genes related to intracellular pathways and regulators of gene expression (9).
In summary, this large body of evidence supports the concept that prolonged GC exposure modulates the epigenetic landscape across the genome by inducing DMR and histone modifications. Some epigenetic modifications are persistent, and this may partially explain the incomplete reversibility of some of CS features following clinical remission.
Glucocorticoid-Target Genes in Cushing Syndrome
A detailed identification and characterization of GC-target genes may shed light in the understanding of the pathophysiology and treatment response in patients with CS. For instance, the GC regulation of pro-opiomelanocortin (POMC) expression as part of the physiologic GC negative feedback may be impaired in Cushing disease (CD), which is an important mechanism for the maintenance of high GC levels (53). Another example is the interaction between GC and clock genes, which may interfere in the loss of the GC circadian rhythm and may contribute to metabolic disorders in CS (54). Furthermore, the suppressive action of GC on drug targets, such as the somatostatin receptor (subtype 2), may influence the efficacy of first-generation somatostatin receptor ligands in normalizing cortisol levels in CD (55). Here we describe how GCs using epigenetic machinery influence the expression of important target genes and their implications in CS.
FKBP5
FK506 binding protein (FKBP5) plays an important role in the regulation of hypothalamic-pituitary-adrenal (HPA) system (56). As part of the GC negative feedback loop, GC binds to hypothalamic and pituitary GR. In the cytoplasm, GR is bound to a multi-protein complex including FKBP5. FKBP5 modulates GR action by decreasing GR binding affinity to GC and by preventing GR translocation from cytoplasm to nucleus (57, 58). In other words, an increase of FKBP5 expression is inversely correlated with GR activity and results in GC resistance leading to an impaired negative feedback regulation in the HPA axis (59).
FKBP5 is a GC-responsive gene; its upregulation by GC is part of an intracellular negative short-feedback loop (60). The mechanism by which GC regulates FKBP5 expression was shown to include inhibition of DNA methylation (44). In a model for CS, mice treated with corticosterone for 4 weeks had a reduced level of DNA methylation of FKBP5 in DNA extracted from whole blood, which was strongly correlated in a negative manner with GC concentration. Interestingly, a negative correlation was also observed between the degree of FKBP5 gene methylation measured at 4 weeks of GC exposure and the percentage of mice visceral fat (61). Accordingly, previous studies have provided compelling evidence of decreased methylation in the FKBP5 gene in patients with active CS compared to healthy control (10, 46). Even in patients with CS in remission, previous data have suggested a small decrease in FKBP5 methylation levels compared to healthy controls (9, 10). In an in vitro study, it was demonstrated that, by decreasing DNMT1 expression, GC is able to reduce FKBP5 methylation levels and, therefore, increase its expression (44).
Likewise, FKBP5 mRNA is also sensitive to GC exposure. A time-dependent increase in blood FKBP5 mRNA after single-dose prednisone administration has been demonstrated in healthy humans (62). Accordingly, patients with ACTH-dependent CS had higher blood FKBP5 mRNA levels compared with healthy controls, and after a successful surgery, FKBP5 mRNA returned to baseline levels (63). Furthermore, in another study, blood FKBP5 mRNA was inversely correlated with FKBP5 promoter methylation and positively correlated with 24-hour urine free cortisol (UFC) levels in patients with CS (46). Taken together, this fine-tuning of FKBP5 DNA methylation and mRNA according to the level of GC suggests that FKBP5 can be used as a biomarker to infer the magnitude of GC exposure.
POMC and Corticotropin-Releasing Hormone
The partial resistance of the corticotroph adenoma to GC negative feedback is a hallmark of CD. Indeed, the lack of this inhibitory effect constitutes a method to diagnose CD, that is, with the dexamethasone suppression test. One of the mechanisms related to the insensitivity to GC can be attributed to GR mutations which are, however, rarely found in corticotrophinomas (64). Another mechanism that was uncovered in corticotroph adenomas is an overexpression of the HSP90 chaperone resulting in reduced affinity of GR to its ligand and consequently GR resistance (53, 65).
In addition, the loss of protein expression of either Brg1, ATPase component of the SWI/SNF chromatin remodeling complex, or HDAC2 has been linked to GC resistance in about 50% of some adenomas (66). The trans-repression process on POMC transcription achieved by GC involves both the histone deacetylation enzyme and Brg1. One mechanism of corticotropin-releasing hormone (CRH)-induced POMC expression is through an orphan nuclear receptor (NR) related to NGFI-B (Nur77). NGFI-B binds to the NurRE sequence in the promoter region of POMC gene and recruits a co-activator to mediate its transcription. In a tethering mechanism, the GR directly interacts with NGFI-B to form a trans-repression complex, which contains the GR itself, Brg1, the nuclear receptor, and HDAC2; the latter being essential to block the gene expression through chromatin remodeling process (53, 66).
In CD, hypercortisolism exerts a negative feedback at CRH secretion from the hypothalamus (67). The mechanism involved in GR-induced suppression of CRH expression is through direct binding to a nGRE in the promoter region of CRH gene and subsequent recruitment of repressor complexes. In a rat hypothalamic cell line, it was demonstrated that Dex-induced CRH repression occurs through coordinated actions of corepressors involving Methyl-CpG-binding protein 2 (MeCP2), HDAC1, and DNA methyltransferase 3B (DNMT3B). Possibly, GR bound to nGRE recruits DNMT3B to the promoter in order to methylate a specific region, subsequently binding MeCP2 on these methylated sites followed by the recruitment of chromatin modify corepressor HDAC1, ultimately resulting in CRH suppression. Another possibility is that 2 independent complexes, one consisting of GR with DNMT3 for the methylation and the other the MeCP2, bound to methylated region, interact with HDAC1 to induce repression (68).
Clock Genes
The clock system and the HPA axis are interconnected regulatory systems. Cortisol circadian rhythm is modulated by the interaction between a central pacemaker, located in the hypothalamic suprachiasmatic nuclei, and the HPA axis (69). At the molecular level, mediators of the clock system and cortisol also communicate with each other, both acting as transcription factors of many genes to influence cellular functions.
In CS, the impact of chronic GC exposure on clock genes expression was recently evaluated using peripheral blood samples from patients with active disease compared with healthy subjects. The circadian rhythm of peripheral clock gene expression (CLOCK, BMAL, PER1-3, and CRY1) was abolished as a result of hypercortisolism, and that may contribute to metabolic disorders observed in Cushing patients (70). Another study, which investigated persistent changes induced by hypercortisolism in visceral adipose tissue, found that the expression of clock genes, such as PER1, remained altered in association with persistent epigenetic changes in both H3K4me3 and H3K27ac induced by hypercortisolism even after the resolution of hypercortisolism (12). This suggests that chronic exposure to GC may induce sustained epigenetic changes that can influence clock genes expression. Nevertheless, further studies are warranted to better elucidate how long-term exposure to GC impacts clock genes expression using the epigenetic machinery.
Glucocorticoid Effects on MicroRNAs
Along with histone modification and DNA methylation, microRNAs (miRNAs) have emerged as an epigenetic mechanism capable of impacting gene expression without changing DNA sequence (15). Interestingly, miRNA expression itself is also under the influence of epigenetic modifications through promoter methylation like any other protein-encoding genes (71).
MicroRNAs are small (about 20-25 nucleotides in length) non-coding RNAs that are important in transcriptional silencing of messenger RNA (mRNA). By partially pairing with mRNA, miRNAs can either induce mRNA degradation or inhibit mRNA translation to protein. MiRNAs regulate the translation of about 50% of the transcriptome, allowing them to play an important role in a wide range of biological functions, such as cell differentiation, proliferation, metabolism, and apoptosis under normal physiological and pathological situations. Some miRNAs can be classified as oncogenes or tumor suppressing genes, and aberrant expression of miRNAs may be implicated in tumor pathogenesis (71-73).
Insight into the regulation of miRNA expression is, therefore, crucial for a better understanding of tumor development and other human diseases, including cardiac, metabolic, and neurological disorders (73, 74). There are different regulatory mechanisms involved in miRNA expression, including transcriptional factors such as GR-GC. GC may modulate miRNA expression through direct binding to GRE in the promoter region of the host gene, as observed in hemopoietic tumor cells (75). In addition to transcriptional activation, in vascular smooth muscle cells, Dex treatment induces downregulation of DNMT1 and DNMT3a protein levels and reduces the methylation of miRNA-29c promoter, resulting in an increased expression of miRNA-29c (76). Interestingly, it was demonstrated that the increased expression of miRNA-29 family (miRNA-29a, -29b, and -29c) associates with metabolic dysfunction, such as obesity and insulin resistance, which pertains to CS (77, 78). With regard to metabolic dysfunction, miRNA-379 expression was shown to be upregulated by GC and its overexpression in the liver resulted in elevated levels of serum triglycerides associated with very low-density lipoprotein (VLDL) fraction in mice (79). In obese patients, the level of hepatic miRNA-379 expression was higher compared to nonobese patients and positively correlated with serum cortisol and triglycerides (79). Hence, GC-responsive miRNA may be, at least in part, a mediator to GC-driven metabolic conditions in CS.
In pathological conditions, such as seen in CS, prolonged exposure to an elevated cortisol level results in a wide range of comorbidities. It can be hypothesized that the chronic and excessive glucocorticoid levels may induce an aberrant miRNA expression that might impact several cellular processes related to bone and cardiometabolic disorders. A recent study addressed the impact of hypercortisolism on bone miRNA of patients with active CD compared to patients with nonfunctional pituitary adenomas. Significant changes in bone miRNA expression levels were observed, suggesting that the disruption of miRNA may be partially responsible for reduced bone formation and osteoblastogenesis (80). Similarly, altered expression levels of selected miRNAs related to endothelial biology in patients with CS may point to a contribution to a high incidence of cardiovascular disorders in Cushing patients (81). Therefore, dysregulated miRNAs as a consequence of high cortisol levels may underpin the development and progression of comorbidities related to CS. To the best of our knowledge, it is currently not clear whether miRNA dysregulation persists after resolution of hypercortisolism, thus contributing to the persistence of some comorbidities. This hypothesis needs to be further investigated.
MicroRNA can also be used as a diagnostic tool in CS. A study was performed to identify circulating miRNA as a biomarker to differentiate patients with CS from patients with suspected CS who had failed diagnostic tests (the control group) (82). It was observed that miRNA182-5p was differentially expressed in the CS cohort compared to the control group; therefore, it may be used as a biomarker (82). However, a large cohort is necessary to validate this finding (82). In corticotroph tumors, downregulation of miRNA 16-1 expression was observed relative to normal pituitary tissue (83). In contrast, the plasma level of miRNA16-5p was found to be significantly higher in CD compared to ectopic Cushing (EAS) and healthy controls (84). This finding suggests that miRNA16-5p may be a biomarker capable to differentiate the 2 forms of ACTH-dependent Cushing (84).
Epidrugs and Glucocorticoid Action in Cushing's Syndrome
The interest in understanding the epigenetic mechanism of GC action in the context of CS is based on reversibility of epi-marks, such as DNA methylation and histone modifications, using epidrugs (85, 86). The biological characteristics of epigenetic drugs and their target have been extensively explored. Their effectiveness as antitumor drugs have been tested on corticotroph tumors using in vitro studies (87-89). However, a limited number of studies have explored the role of epidrugs as a therapeutic tool in reversing the genomic action of GC in CS, particularly in comorbidities induced by hypercortisolism (90, 91).
The use of histone deacetylase inhibitors (HDACi) may reduce the genomic action of GC (90-92). It has been demonstrated that the use of the HDAC inhibitor valproic acid increases the acetylation level of GR, consequently attenuating the genomic action of GC. In an experimental Cushing model in rats, the use of valproic acid decreased expression of genes related to lipogenesis, gluconeogenesis, and ion regulators in the kidney that ultimately reduces hepatic steatosis, hyperglycemia, and hypertension in ACTH-infused rats (90, 91).
More studies evaluating the effects of epidrugs influencing the GC actions are warranted to further elucidate the underlying mechanisms and to explore potential treatment modalities to reverse long-lasting consequences of chronic corticoid exposure.
Conclusions
In physiologic conditions, GC are secreted in pulses following a circadian rhythm pattern, as opposed to a constant, chronic, and high GC exposure in CS. This pathological pattern may account for numerous devastating effects observed in CS (7). Yet, the expressed genome in response to chronic GC exposure may potentially be abnormal, leading to dysregulation in clock genes, among other effects.
GC levels may return to a normal circadian pattern in response to a successful treatment, but with incomplete reversibility of some CS features, which may in part be explained by epigenetic changes. The epigenetic machinery is used by GC to induce dynamic changes in chromatin to modulate gene expression. (Fig. 2) It seems that most of chromatin modifications are reversible, but some may persist resulting in long-term epigenetic changes. (Table 1)
Table 1.Evidence of interaction between glucocorticoid and epigenetic machinery
Epigenetic changes/epigenetic enzymes Action Histone acetylation (HAT) Histone deacetylation (HDAC) -
GR recruit histone deacetylases (HDACs) to turn chromatin less accessible and suppress gene transcription (25, 35).
-
The trans-repression process on POMC transcription achieved by glucocorticoids (GC) involves the histone deacetylation enzyme (HDAC2).
-
GC mediates the upregulation of HDAC2 in rats exposed to chronic stress (40).
Histone demethylase (JMJD3) -
GC suppress transcription of JMJD3 in endothelial cells treated with TNFα (41).
Histone modifications -
Using ChIP-seq, a study in mice treated for 5 weeks with corticosterone showed higher levels of histone modifications (H3K4me3, H3K27ac) compared to control mice. In mice after a 10-week washout period, persistence of this epigenetic fingerprint was observed, which was associated with long-lasting changes in gene expression (12).
DNA methylation (DNMT3B) and histone deacetylation (HDAC1) -
GC mediates CRH downregulation through DNMT3B to the promoter in order to methylate a specific region and recruitment of chromatin modify corepressor HDAC (68).
DNA hypomethylation -
GC induces downregulation of DNMT1 in AtT20 (mouse corticotroph adenoma cell line) (20).
-
GC induces upregulation of TET enzyme expression which was described in retinal and osteocyte cell line model (42, 43).
-
An experimental study in mice previously exposed to high levels of GC showed differentially methylated regions (DMR) induced by GC treatment, of which the majority was loss of the methylation (11).
-
Reduced DNA methylation in FKBP5 gene was found in patients in active disease and also in remission state of Cushing syndrome (CS) as compared to a healthy control group (10).
-
A genome-wide DNA methylation analysis showed a lower average of DNA methylation in patients in remission of CS compared to controls (9).
-
A study using whole blood methylation profile demonstrated an association between cortisol excess and DNA hypomethylation in patients with CS (46).
Further studies are needed to elucidate how chronic exposure to GC leads to incomplete reversibility of CS morbidities via sustained modulation of the epigenetic machinery and possibly other mechanisms. Subsequent identification of therapeutic targets may offer new perspective for treatments, for example, with epidrugs, aiming to reverse hypercortisolism-related comorbidities.
Funding
The authors received no financial support for this manuscript.
Disclosures
T.P., R.A.F., and L.J.H. have nothing to declare.
Data Availability
Data sharing is not applicable to this article, as no datasets were generated or analyzed during the current study.
-
 1
1
-
-
Wow - it's nearly 18 years since my cancer surgery.
So, I've been back on the growth hormone for about 8 years. I still don't feel like it's doing much/any good but it must be since tests come out ok, my endo still prescribes it and my insurance still pays for a huge chunk of it.
~~~
Cushing's awareness discussions come out in the oddest ways.
Last night, we were out at a Lebanese restaurant couple with a young couple from India and were talking about foods we liked. I said that I liked spicy foods in general.
That led to the fact that I haven't had a good sense of smell since my transsphenoidal surgery and that things taste better when I can smell them. That got us around to why I'd had the surgery and what the symptoms of Cushing's are.
Our dinner companions will probably never hear of Cushing's again but if they do, they'll know what it is!

-
-
Abstract
Cushing’s syndrome is a constellation of features occurring due to high blood cortisol levels. We report a case of a 47-year-old male with a history of recurrent olfactory neuroblastoma (ONB). He presented with bilateral lower limb weakness and anosmia and was found to have Cushing’s syndrome due to high adrenocorticotropic hormone (ACTH) levels from an ectopic source, ONB in this case. Serum cortisol and ACTH levels declined after tumor removal.
Introduction
Olfactory neuroblastoma (ONB), or esthesioneuroblastoma, is a rare malignancy arising from neuroepithelium in the upper nasal cavity. It represents approximately 2% of all nasal passage tumors, with an incidence of approximately 0.4 per 2.5 million individuals [1]. ONB shares similar histological features with small round blue cell neoplasms of the nose. Ectopic hormone secretion is a very rare feature associated with these tumors. Five-year overall survival is reported to be between 60% and 80% [2,3]. The age distribution is either in the fifth to sixth decade of life [4,5], or in the second and sixth decades [6].
Features of Cushing’s syndrome (moon face, buffalo hump, central obesity hypertension, fragile skin, easy bruising, fatigue, muscle weakness) are due to high blood cortisol levels [7]. It can be either primary (cortisol-secreting adrenal tumor), secondary (adrenocorticotropic hormone (ACTH)-secreting pituitary tumor, also called Cushing disease), or ectopic ACTH secretion (from a non-pituitary source). All three types share similar features [8].
Ectopic ACTH syndrome (EAS) is due to an extra pituitary tumor, producing ACTH. It accounts for 12-17% of Cushing's syndrome cases [9]. Most cases of EAS-producing tumors are in the lungs, mediastinum, neuroendocrine tumors of the gastrointestinal tract, and pheochromocytomas [9]. Ectopic ACTH secretion from an ONB is very rare. As of 2015, only 18 cases were reported in the literature [10]. Here, we report such a case.
Case Presentation
Our patient is a 47-year-old Bangladeshi male, with a history of recurrent ONB that was resected twice in the past (transsphenoidal resection in 2016 and 2019) with adjuvant radiotherapy, no chemotherapy was given. He also had diabetes mellitus type 1 (poorly controlled) and hypertension. He presented with bilateral lower limb weakness, anosmia, decreased oral intake, loss of taste for one week, and bilateral submandibular swelling that increased in size gradually over the past two years. There was no history of fever, cough, abdominal pain, or exposure to sick contacts. The patient reported past episodes of similar symptoms, but details are unclear. The patient's family history is positive for diabetes mellitus type 1 in both parents. Lab tests in the emergency department showed hypokalemia and hyperglycemia as detailed in Table 1. He was admitted for further workup of the above complaints.
Test Patient Results Reference Range Unit Status Hemoglobin 14.7 13-17 g/dL Normal White blood cell (WBC) 17.9 4-10 10*9/L High Neutrophils 15.89 2-7 10*9/L High Lymphocytes 1.07 1-3 10*9/L Normal Sodium 141 136-145 mmol/L Normal Potassium 2.49 3.5-5.1 mmol/L Low (Panic) Chloride 95 98-107 mmol/L Low Glucose 6.52 4.11-5.89 mmol/L Elevated C-reactive protein (CRP) 0.64 Less than 5 mg/L Normal Erythrocyte sedimentation rate (ESR) 2 0-30 mm/h Normal Creatinine 73 62-106 µmol/L Normal Uric acid 197 202.3-416.5 µmol/L Normal Alanine aminotransferase (ALT) 33.2 0-41 U/L Normal Aspartate aminotransferase (AST) 18.6 0-40 U/L Normal International Normalised Ratio (INR) 1.21 0.8-1.2 sec High Prothrombin time (PT) 15.7 12.3-14.7 sec High Lactate dehydrogenase (LDH) 491 135-225 U/L High Thyroid-stimulating hormone (TSH) 0.222 0.27-4.20 mIU/L Low Adrenocorticotropic hormone (ACTH) 106 ≤50 ng/L Elevated Cortisol (after dexamethasone suppression) 1750 Morning hours (6-10 am): 172-497 nmol, Afternoon hours (4-8 pm): 74.1-286 nmol nmol/L Elevated (failure of suppression) 24-hour urine cortisol (after dexamethasone suppression) 5959.1 <120 nmol/24 hrs nmol/24hr Elevated (failure of suppression) Table 1: Results of blood test at the time of hospitalization. Hypokalemia and high values of adrenocorticotropic hormone and cortisol were confirmed.
On examination, the patient's vital signs were as follows: blood pressure was 154/77 mmHg, heart rate of 60 beats per minute, respiratory rate was 18 breaths per minute, oxygen saturation of 98% on room air, and a temperature of 36.7°C. The patient had a typical Cushingoid appearance with a moon face, buffalo hump, purple striae on the abdomen, central obesity, and hyperpigmentation of the skin. Submandibular lymph nodes were enlarged bilaterally. The examination of the submandibular lymph nodes showed a firm, fixed mass extending from the angle of the mandible to the submental space on the left side. Neurological examination showed weakness in both legs bilaterally (strength 3/5) and anosmia (checked by orthonasal smell test). The rest of the neurological exam was normal.
Laboratory findings revealed (in Table 1) a marked hypokalemia of 2.49 mmol/L and hyperglycemia of 6.52 mmol/L. The serum cortisol level was elevated at 1587 nmol/L. Serum ACTH levels were raised at 106 ng/L (normal value ≤50 ng/L). Moreover, the high-dose dexamethasone suppression test failed to lower the serum ACTH levels and serum and urine cortisol. Serum cortisol level after the suppression test was 1750 nmol/L, while 24-hour urine cortisol after the test was 5959.1 nmol/24hr. Serum ACTH levels after the test also remained high at 100mg/L. This indicated failure of ACTH suppression by high-dose dexamethasone, which points towards ectopic ACTH production. Other blood tests (complete blood count, liver function tests) were insignificant.
A computed tomography scan with contrast (CT scan) of the chest, abdomen, and pelvis, with a special focus on the adrenals, was negative for any malignancy or masses. CT scan of the neck showed bilaterally enlarged submandibular lymph nodes and an enlarged right lobe of the thyroid with nodules. Fine needle aspiration (FNA) of the thyroid nodules revealed a benign nature. Magnetic resonance imaging (MRI) of the brain showed a contrast-enhancing soft tissue lesion (18x18x10mm) in the midline olfactory groove area with extension into the frontal dura and superior sagittal sinus, suggesting recurrence of the previous ONB. There was evidence of previous surgery also. The pituitary gland was normal (Figures 1-2).
Figure 1: A brain MRI (T1-weighted; without contrast; sagittal plane) shows a soft tissue lesion located in the midline olfactory groove area. Dural surface with extension into anterior frontal dura.
MRI: Magnetic resonance imaging
Figure 2: A brain MRI (T2-weighted; without contrast; axial plane) shows a soft tissue lesion located in the midline olfactory groove area.
MRI: Magnetic resonance imaging
Octreotide scintigraphy showed three focal abnormal uptakes in the submandibular cervical nodes. Additionally, there was a moderate abnormal uptake at the midline olfactory groove with bilateral extension (Figure 3).
Figure 3: Whole-body octreotide scan (15 mCi 99mTc-Octreotide IV) demonstrates three focal abnormal uptakes: the largest (5.2 x 2.4 cm) in the left submandibular region, and two smaller ones on the right, suggestive of lymph node uptake. Additional abnormal uptake was seen along the midline of the olfactory groove region with bilateral extension. No other significant abnormal uptake was identified.
On microscopic examination, an excisional biopsy after the transcranial resection surgery of the frontal skull base tumor showed nests and lobules of round to oval cells with clear cytoplasm, separated by vascular and hyalinized fibrous stroma (Figures 4A-4B). Tumor cells show mild to moderate nuclear pleomorphism, and fine chromatin (Figure 4C). A fibrillary neural matrix is also present. Some mitotic figures can be seen. Immunohistochemical stains revealed positive staining for synaptophysin (Figure 4D) and chromogranin (Figure 4E). Stains for CK (AE1/AE3), CD45, Desmin, and Myogenin are negative. Immunostaining for ACTH was focally positive (Figure 4F), while the specimen of the cervical lymph nodes showed the same staining, indicating metastases. The cytomorphologic and immunophenotypic features observed are consistent with a Hyams grade II ONB, with ectopic ACTH production.
Figure 4: Histopathological and immunohistochemical findings of olfactory neuroblastoma.
A (100x magnification) and B (200x magnification) - hematoxylin and eosin (H-E) staining shows cellular nests of round blue cells separated by hyalinized stroma. C (400x magnification) - nuclei show mild to moderate pleomorphism with fine chromatin. D (100x magnification) - an immunohistochemical stain for synaptophysin shows diffuse, strong cytoplasmic positivity within tumor cells. E (200x magnification) - tumor cells are positive for chromogranin. F (400x magnification) - ACTH cytoplasmic expression in tumor cells.
ACTH: adrenocorticotropic hormone
For his resistant hypokalemia, he had to be given intravenous (IV) and oral potassium chloride (KCL) repeatedly. The patient underwent transcranial resection of the frontal skull base tumor. The patient received cefazolin for seven days, and hydrocortisone for four days. After transcranial resection, his cortisol level decreased to 700 nmol/L. Furthermore, ACTH dropped, and serum potassium also normalized. Subsequently, the patient was transferred to the intensive care unit (ICU) for meticulous monitoring and continued care. In the ICU, the patient developed one episode of a generalized tonic-clonic seizure, which aborted spontaneously, and the patient received phenytoin and levetiracetam to prevent other episodes. A right-sided internal jugular vein and left transverse sinus thrombosis were also developed and treated with enoxaparin sodium. Following surgery, his low potassium levels improved, resulting in an improvement in his limb weakness. His other symptoms also gradually improved after surgery. Three weeks following the primary tumor resection, he underwent bilateral neck dissection with right hemithyroidectomy, for removal of the metastases. The patient opted out of chemotherapy and planned for an international transfer to his home country for further management. Other treatments that he received during hospitalization were ceftriaxone, azithromycin, and Augmentin®. Insulin was used to manage his diabetes, perindopril to regulate his blood pressure, and spironolactone to increase potassium retention. Omeprazole was administered to prevent GI bleeding and heartburn/gastroesophageal reflux disease relief after discharge.
Discussion
ONB was first described in 1924, and it is a rare neuroectodermal tumor that accounts for 2% of tumors affecting the nasal cavity [11]. Even though ONB has a good survival rate, long-term follow-up is necessary due to the disease's high recurrence rate [2]. ONB recurrence has been approximated to range between 30% and 60% after successful treatment of the primary tumor [12]. Recurrent disease is usually locoregional and tends to have a long interval to relapse with a mean of six years [12]. The first reported case of ectopic ACTH syndrome caused by ONB was in 1987 by M Reznik et al., who reported a 48-year-old woman with ONB who developed a Cushing-like syndrome 28 months before her death [13].
The occurrence of Cushing’s syndrome due to ectopic ACTH can occur either in the initial tumor or even years later during its course or after recurrence [3,6,9,14]. Similar to the case of Abe et al. [3], our patient also presented with muscle weakness due to hypokalemia, which is a feature of Cushing’s syndrome. Hypokalemia is present at diagnosis in 64% to 86% of cases of EAS and is resistant to treatment [9,14], as seen in our case. In our patient, the exact time of development of Cushing’s syndrome could not be ascertained due to the non-availability of previous records. However, according to the patient, he started developing abdominal obesity, pigmentation, and buffalo hump in 2021 about two years after his second surgery for ONB.
The distinction between pituitary ACTH and ectopic ACTH involves utilizing CT/MRI of the pituitary, corticotropin-releasing hormone (CRH) stimulation test with petrosal sinus blood sampling, high dose dexamethasone suppression test, and checking serum K+ (more commonly low in ectopic ACTH) [2,15,16]. In our case, a CRH stimulation test was not available but CT/MRI brain, dexamethasone test, low serum potassium, plus the postoperative fall in cortisol levels, all pointed towards an ectopic ACTH source.
Conclusions
In conclusion, this case highlights the rare association between ONB and ectopic ACTH syndrome, which developed after tumor recurrence. The patient's unique presentation of bilateral lower limb weakness and hypokalemia can cause diagnostic challenges, emphasizing the need for comprehensive diagnostic measures. Surgical intervention proved crucial, with postoperative cortisol values becoming normal, highlighting the efficacy of this approach. The occurrence of ectopic ACTH production in ONB patients, although very rare, is emphasized, so that healthcare professionals who deal with these tumors are aware of this complication. This report contributes valuable insights shedding light on the unique ONB manifestation causing ectopic ACTH syndrome. The ongoing monitoring of the patient's clinical features will further enrich the understanding of the course of this uncommon phenomenon in the medical literature.
References
- Thompson LD: Olfactory neuroblastoma. Head Neck Pathol. 2009, 3:252-9. 10.1007/s12105-009-0125-2
- Abdelmeguid AS: Olfactory neuroblastoma. Curr Oncol Rep. 2018, 20:7. 10.1007/s11912-018-0661-6
- Abe H, Suwanai H, Kambara N, et al.: A rare case of ectopic adrenocorticotropic hormone syndrome with recurrent olfactory neuroblastoma. Intern Med. 2021, 60:105-9. 10.2169/internalmedicine.2897-19
- Yin Z, Wang Y, Wu Y, et al.: Age distribution and age-related outcomes of olfactory neuroblastoma: a population-based analysis. Cancer Manag Res. 2018, 10:1359-64. 10.2147/CMAR.S151945
- Platek ME, Merzianu M, Mashtare TL, Popat SR, Rigual NR, Warren GW, Singh AK: Improved survival following surgery and radiation therapy for olfactory neuroblastoma: analysis of the SEER database. Radiat Oncol. 2011, 6:41. 10.1186/1748-717X-6-41
- Elkon D, Hightower SI, Lim ML, Cantrell RW, Constable WC: Esthesioneuroblastoma. Cancer. 1979, 44:3-1087. 10.1002/1097-0142(197909)44:3<1087::aid-cncr2820440343>3.0.co;2-a
- Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM: The diagnosis of Cushing's syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008, 93:1526-40. 10.1210/jc.2008-0125
- Chabre O: Cushing syndrome: physiopathology, etiology and principles of therapy [Article in French]. Presse Med. 2014, 43:376-92. 10.1016/j.lpm.2014.02.001
- Isidori AM, Lenzi A: Ectopic ACTH syndrome. Arq Bras Endocrinol Metabol. 2007, 51:1217-25. 10.1590/s0004-27302007000800007
- Kunc M, Gabrych A, Czapiewski P, Sworczak K: Paraneoplastic syndromes in olfactory neuroblastoma. Contemp Oncol (Pozn). 2015, 19:6-16. 10.5114/wo.2015.46283
- Finlay JB, Abi Hachem R, Jang DW, Osazuwa-Peters N, Goldstein BJ: Deconstructing olfactory epithelium developmental pathways in olfactory neuroblastoma. Cancer Res Commun. 2023, 3:980-90. 10.1158/2767-9764.CRC-23-0013
- Ni G, Pinheiro-Neto CD, Iyoha E, et al.: Recurrent esthesioneuroblastoma: long-term outcomes of salvage therapy. Cancers (Basel). 2023, 15:1506. 10.3390/cancers15051506
- Reznik M, Melon J, Lambricht M, Kaschten B, Beckers A: Neuroendocrine tumor of the nasal cavity (esthesioneuroblastoma). Apropos of a case with paraneoplastic Cushing's syndrome [Article in French]. Ann Pathol. 1987, 7:137-42.
- Kadoya M, Kurajoh M, Miyoshi A, et al.: Ectopic adrenocorticotropic hormone syndrome associated with olfactory neuroblastoma: acquirement of adrenocorticotropic hormone expression during disease course as shown by serial immunohistochemistry examinations. J Int Med Res. 2018, 46:4760-8. 10.1177/0300060517754026
- Clotman K, Twickler MTB, Dirinck E, et al.: An endocrine picture in disguise: a progressive olfactory neuroblastoma complicated with ectopic Cushing syndrome. AACE Clin Case Rep. 2017, 3:278-83. 10.4158/EP161729.CR
- Chung YS, Na M, Ku CR, Kim SH, Kim EH: Adrenocorticotropic hormone-secreting esthesioneuroblastoma with ectopic Cushing’s syndrome. Yonsei Med J. 2020, 61:257-61. 10.3349/ymj.2020.61.3.257
-
 1
1
-
You know, I'm still not sure if the GH is worth it, even after almost 20 years (with some hiccups). I had to stop after I've had kidney cancer and flying with the stuff is such a pain due to the refrigeration (more about that here).
One time we were going on a cruise out of New York so we were visiting our son first. I was in a hotel and I put the whole case in an ice bucket. The ice melted... I wasn't sure if the water had seeped into the injector pen or not so I threw it away and went on the cruise without it. I never noticed any bad effects from the week or so with no GH.
So, last January when we went on another cruise, I didn't bother to take it with no ill effects.
So, it doesn't seem like it's working for me but my endo (Dr. Salvatori at Johns Hopkins) is happy so I guess my non-schedule is ok. Maybe I'll just die sooner than I would have.

-
 1
1
-
-
Authors Stasiak M , Witek P, Adamska-Fita E, Lewiński A
Received 27 December 2023
Accepted for publication 20 March 2024
Published 8 April 2024 Volume 2024:16 Pages 35—42
DOI https://doi.org/10.2147/DHPS.S453105
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Hemalkumar B Mehta
Magdalena Stasiak,1 Przemysław Witek,2 Emilia Adamska-Fita,1 Andrzej Lewiński1,3
1Department of Endocrinology and Metabolic Diseases, Polish Mother’s Memorial Hospital—Research Institute, Lodz, Poland; 2Department of Internal Medicine, Endocrinology and Diabetes, Medical University of Warsaw; Mazovian Brodnowski Hospital, Warszawa, Poland; 3Department of Endocrinology and Metabolic Diseases, Medical University of Lodz, Lodz, Poland
Correspondence: Magdalena Stasiak, Department of Endocrinology and Metabolic Diseases, Polish Mother’s Memorial Hospital—Research Institute, 281/289 Rzgowska Street, Lodz, 93-338, Poland, Tel +48502049292, Fax +48422711140, Email mstasiak33@gmail.com
Abstract: Cushing’s disease (CD) is the most common cause of endogenous hypercortisolism. Osilodrostat was demonstrated to be efficient in treating CD, and the mean average dose required for CD control was < 11 mg/day. Potential differences in osilodrostat treatment between cortisol-producing adenoma (CPA) and CD have not been reported. The aim of this study was to present two patients with CPA in whom significant differences in the response to therapy compared to CD were found. We demonstrated a case of inverse response of cortisol levels with adrenal tumor progression during the initial dose escalation (Case 1). Simultaneously, severe exaggeration of hypercortisolism symptoms and life-threatening hypokalemia occurred. A further rapid dose increase resulted in the first noticeable cortisol response at a dose of 20 mg/day, and a full response at a dose of 45 mg/day. We also present a case that was initially resistant to therapy (Case 2). The doses required to achieve the first response and the full response were the same as those for Case 1. Our study demonstrated that osilodrostat therapy in patients with CPA may require a different approach than that in CD, with higher doses, faster dose escalation, and a possible initial inverse response or lack of response.
Keywords: osilodrostat, adrenal adenoma, hypercortisolism, ACTH-independent, adverse events, hypokalemiaIntroduction
Chronic persistent hypercortisolism is a life-threatening condition that requires effective treatment. Untreated exposure to excessive cortisol secretion leads to severely increased morbidity and mortality due to cardiovascular diseases, thromboembolic events, sepsis, visceral obesity, impairment of glucose metabolism, and dyslipidaea, as well as musculoskeletal disorders, such as myopathy, osteoporosis, and skeletal fractures. Moreover, neuropsychiatric disorders, such as impairment of cognitive function, depression, or mania, as well as impairment of reproductive function can frequently occur.1,2 Cushing’s disease (CD) – a disorder caused by a pituitary adenoma secreting adrenocorticotropic hormone (ACTH) – is the most common cause of hypercortisolism. Cushing’s syndrome (CS) includes all other causes of cortisol excess, including ectopic ACTH production as well as direct cortisol overproduction by adrenal adenoma (cortisol-producing adenoma [CPA]) or adrenocortical carcinoma (ACC). Approximately 10% of hypercortisolism cases result from CPA. The first line therapy is a surgical resection of the tumor, which is the source of hormone excess. However, in many patients surgery is not fully efficient and other therapies are required to reduce cortisol levels. Additionally, due to severe cardiovascular complications and unstable DM, the surgical approach sometimes entails unacceptable risk and it is frequently postponed until cortisol levels are lowered. Pharmacotherapy with steroidogenesis inhibitors reduces cortisol levels and improves the symptoms of hypercortisolism.1,2 As CD is the most common cause of cortisol excess, most studies have focused on the efficacy and safety of novel steroidogenesis inhibitors, including patients with CD only.3–6 This is exactly the case with osilodrostat – a new potent inhibitor of 11β-hydroxylase.3–6 More data are available for metyrapone efficacy and safety in CSA,7 as the drug has been available much longer than osilodrostat. A study by Detomas et al, which reported results of comparison of efficacy of metyrapone and osilodrostat, included 4 patients with adrenal CS, among whom one CPA patient was treated with osilodrostat.8 Osilodrostat is approved in the United States to treat CD in patients in whom pituitary surgery was not curative or is contraindicated.9 In Poland, osilodrostat therapy is available for patients with all kinds of endogenous hypercortisolism not curative with other approaches, within a national program of emergency access to drug technologies.10 Reports on osilodrostat application in CPA are highly valuable as data on potential differences in the treatment regimens between CD and CPA are scarce.
Here, we present two patients with CPA in whom the response and doses of osilodrostat were different from those reported in patients with CD. The main purpose of this study was to demonstrate that the efficacy of osilodrostat in CPA is high, although initial resistance to treatment or even deterioration of hypercortisolism can occur during the application of lower doses of the drug.
Materials and Methods
Study Design and Patients
We retrospectively analyzed medical files of two consecutive patients with CPA treated with osilodrostat. The analysis included medical history, laboratory and imaging results as well as a detailed reports of adverse events.
Laboratory and Imaging Procedures
Serum cortisol and ACTH levels were measured by electrochemiluminescence immunoassay (ECLIA) using a Cobas e601 analyzer (Roche Diagnostics, Indianapolis, IN, USA). UFC excretion was measured by chemiluminescent microparticle immunoassay (CMIA) using an Abbott Architect ci4100 analyzer (Abbott, Abbott Park, IL, USA). Cross-reactivity with 11-deoxycortisol for this method is very low (2.1% according to the manufacturer’s data). Potassium levels were measured by ion-selective electrode potentiometry using a Beckman Coulter DxC 700 AU Chemistry Analyzer (Beckman Coulter, Brea, CA, USA). Computed tomography (CT) imaging was performed using a Philips Ingenuity Core 128 system (Philips, the Netherlands).
Ethics Procedures
Informed consent was obtained from all subjects involved in the study. Written informed consent was obtained from the patients for publication of this paper. The approval of Institutional Ethics Committee was obtained to publish the case details (approval code KB 33/2023).
Presentation of the Cases
Case 1
A 51-year-old female was referred to our department in November 2021 because of CPA, disqualified from surgery because of severe hypertension with a poor response to antihypertensive therapy and uncontrolled DM despite high doses of insulin. Additionally, the patient presented with hyperlipidemia and severe obesity (BMI=50.7 kg/m2), gastritis, depression, and osteoarthritis. On admission, she complained of a tendency to gain weight, fragile skin that bruised easily, difficulty with wound healing, susceptibility to infections, and insomnia. Physical examination revealed a moon face with plethora, a buffalo hump, central obesity with proximal muscle atrophy, and purple abdominal striae.
The CPA diagnosis was initially made two years earlier, but the patient did not qualify for surgery due to a hypertensive crisis. Soon after this episode, the SARS-CoV-2 pandemic began, and the patient was afraid of visiting any medical center because her son had died of COVID-19. Therefore, she was referred to our center for life-threatening hypercortisolism two years later.
At the time of admission, computed tomography (CT) imaging revealed a right adrenal tumor of 34x24x37mm, with a basal density of 21 HU and a contrast washout rate typical for adenomas (83%). The size and CT characteristics were identical as they were two years earlier. High serum cortisol levels, undetectable ACTH concentrations, and a lack of physiological diurnal rhythm of cortisol secretion were observed (Table 1). Urinary free cortisol (UFC) excretion was 310 µg/24 h, with an upper normal limit (UNL) of 176 µg/24 h. No cortisol suppression was achieved in high-dose dexamethasone suppression test (DST) (Table 1). Other adrenal-related hormonal parameters were within normal ranges, with values as follows: DHEA-S 42.68 µg/dl, aldosterone 3.24 ng/mL, and renin 59.14 µIU/mL.

Table 1 Laboratory Results Before Osilodrostat Therapy – Case 1
Due to multiple severe systemic complications, including uncontrolled hypertension, decompensated DM, and cardiac insufficiency, treatment with osilodrostat was introduced for life-saving pre-surgical management. Osilodrostat was started at a dose of 1 mg twice daily and gradually increased to 6 mg per day with actually an inverse response of serum cortisol level. The late-night cortisol level increased from 16 µg/dl to 25 µg/dl. As the full effect of the osilodrostat dose can occur even after a few weeks, the patient was discharged from hospital and instructed to contact her attending doctor immediately if any health deterioration was noticed. In the case of improvement in the patient’s condition, the next hospitalization was planned 3 weeks later. After three weeks of no contact with the patient, she was readmitted to our department with life-threatening escalation of hypercortisolism, severe hypokalemia, and further deterioration of hypertension, DM, cardiac insufficiency, dyspnea, and significant edemas, including facial edema. Treatments of hypertension, cardiac insufficiency, and DM were intensified, as presented in Table 2. Despite active potassium supplementation, life-threatening hypokalemia of 2.1 mmol/l occurred. Previously observed depression was exaggerated with severe anxiety and fear of death. The dose of osilodrostat was increased to 8 mg/day, and after three days of treatment a further elevation of serum cortisol was found, with an increase in UFC up to 9 × UNL (1546.2 µg/24 h). Due to an entirely unexpected inverse cortisol response, CT imaging was performed and revealed progression of the adenoma size to 39 × 36 × 40 mm, with a slight increase in density up to 27 HU as compared to the previous CT scan performed a month earlier (Figure 1).

Table 2 Changes in the Most Important Parameters During Osilodrostat Therapy – Case 1
Considering the extremely high risk associated with such a rapid cortisol increase and related complications, decision of fast osilodrostat dose escalation was made. The dose was increased by 5 mg every other day, up to 45 mg per day, and, finally, a gradual decrease in the cortisol level (Table 2) was achieved, with UFC normalization to 168 µg/24 h. During dose escalation, no deterioration in the adverse effects (AEs) of osilodrostat was observed. Conversely, hypokalemia gradually improved despite a simultaneous reduction in potassium supplementation (Table 2). Facial edema decreased and the level of anxiety improved significantly. The course of hypertension severity as well as a summary of the main parameters controlled during treatment and the medications used are presented in Table 2. As soon as the cortisol level normalized, the patient was referred for surgery and underwent right adrenalectomy without any complications. Histopathology results confirmed a benign adenoma of the right adrenal gland (encapsulated, well-circumscribed tumor consisting of lipid-rich cells with small and uniform nuclei, mostly with eosinophilic intracytoplasmic inclusions). After surgery, hydrocortisone replacement therapy was administered. A few days after surgery, blood pressure and glucose levels gradually decreased, and the patient required reduction of antihypertensive and antidiabetic medications. After 22 months of follow-up, the patient’s general condition is good with no signs of recurrence. Antidepressant treatment is no longer required in this patient. Body mass index was significantly reduced to 40 kg/m2. The antihypertensive medication was completely discontinued, and the glucose level is controlled only with metformin. The patient still requires hydrocortisone substitution at a dose of 30 mg/day.
Case 2
A 39-year-old female was referred to our department in November 2022 with a diagnosis of CPA and unstable hypertension, for which surgery was contraindicated. The patient was unsuccessfully treated with triple antihypertensive therapy (telmisartan 40 mg/day, nebivolol 5 mg/day, and lercanidipine 20 mg/day). The patient reported weight gain, muscle weakness, acne, fragile skin that bruised easily, and secondary amenorrhea. Other comorbidities included gastritis, hypercholesterolemia, and osteoporosis. Physical examination revealed typical signs of Cushing’s syndrome, such as abnormal fat distribution, particularly in the abdomen and supraclavicular fossae, proximal muscle atrophy, moon face, and multiple hematomas. A lack of a serum cortisol diurnal rhythm with high late-night serum cortisol and undetectable ACTH levels was found (Table 3). The short DST revealed no cortisol suppression (Table 3), and the UFC result was 725 µg/24 h, which exceeded the UNL more than four times. The serum levels of renin, aldosterone, and 24-h urine fractionated metanephrines were within the normal ranges. Computed tomography imaging revealed a left adrenal gland tumor measuring 25 × 26 × 22 mm, with a basal density of 32 HU and a washout rate typical for adenoma (76%).

Table 3 Laboratory Results Before Osilodrostat Therapy – Case 2
Osilodrostat therapy was administered for preoperative management. The initial daily dose was 2 mg/day, increased gradually by 2 mg every day with no serum cortisol response (late night cortisol levels 15.8–18.5 µg/dl) and no AEs of the drug (Table 4). After the daily dose of osilodrostat reached 10 mg, it was escalated by 5 mg every other day, initially with no serum cortisol reduction. The dose was increased to 45 mg daily (with the lowest detected late-night serum cortisol of 9.6 µg/dl) (Table 4).

Table 4 Changes in the Most Important Parameters During Osilodrostat Therapy – Case 2
After a week of administration of 45 mg daily, UFC normalization was achieved. Despite rapid dose escalation, no AEs were observed during the entire therapy period. Potassium levels were normal without any supplementation (the lowest detected serum potassium level was 3.9 mmol/l; all other results were over 4.0 mmol/l) (Table 4). After UFC normalization, left adrenalectomy was performed without complications. Histopathological examination revealed benign adrenal adenoma. Antihypertensive therapy was reduced only to 2.5 mg of nebivolol daily. The patient’s general condition improved significantly. Currently, hydrocortisone replacement therapy is administered at a dose of 15 mg/day.
Discussion
Osilodrostat is a novel potent steroidogenesis inhibitor whose efficacy and safety have been thoroughly analyzed in clinical trials of patients with CD, the most common cause of endogenous hypercortisolism. No clinical trial of osilodrostat therapy in CPA has been performed, as this disease constitutes only 10% of all cases of endogenous hypercortisolism. Moreover, osilodrostat is not approved by the FDA for hypercortisolism conditions other than CD.9 Therefore, data on potential differences in the treatment regimen are lacking.
During the course of already reported trials in CD, osilodrostat doses were escalated slowly, every 2–3 weeks,3,5,6 with an excellent response to quite low doses of the drug.3–6 In the LINC 2 extension study the median average dose was 10.6 mg/day,5 while in the LINC 3 extension study and the LINC 4 study it was 7.4 mg/day and 6.9 mg/day, respectively.4,6 In most cases, a significant decrease of hypercortisolism was reported with the low doses of osilodrostat (4 or 10 mg/day). Moreover, some patients received 1 mg/day or even 1 mg every other day, with a good response.6 Even in rare cases of CD in whom initial short-term etomidate therapy was given at the beginning of osilodrostat therapy, due to highly severe life-threatening symptoms of hypercortisolism, the final effective dose of osilodrostat was much lower than that in our patients with CPA (25 mg/day vs 45 mg/day) and no increase of cortisol level was observed.11
It should be underlined that many cases of adrenal insufficiency during osilodrostat therapy in patients with CD have been reported,3–6,12,13 and – therefore – low initial dose with slow gradual dose escalation is recommended in patients with CD.1,6,13
In the cases presented here, CPA led to severe hypercortisolism, the complications of which constituted contraindications for surgery. Therefore, osilodrostat therapy was introduced as a presurgical treatment. In Case 1, the therapy was started at low doses according to the approved product characteristics.14 Due to the severity of hypertension, which was uncontrolled despite of active antihypertensive therapy, as well as to unstable DM, the doses were increased faster than recommended. Surprisingly, we immediately observed a gradual increase in hypercortisolism, in both serum cortisol levels and the UFC, with simultaneous burst of complications related to both hypercortisolism itself and 11β-hydroxylase inhibition. Life-threatening episodes of hypertensive crisis responded poorly to standard therapies. Severe exaggeration of cardiac insufficiency could probably be related to these episodes as well as to deep hypokalemia, which occurred despite potassium supplementation. Hypokalemia is a typical complication of treatment with 11β-hydroxylase inhibitors due to the accumulation of adrenal hormone precursors. However, Patient 1 required much higher doses of potassium supplementation, both parenteral and oral, than ever described during osilodrostat therapy.3–6,13 The dose of 20 mg/day of osilodrostat was the first one which led to noticeable cortisol reduction and a decrease in systolic blood pressure (SBP) to below 170 mmHg. Surprisingly, instead of the expected deterioration of hypokalemia, parenteral potassium administration could be stopped with an osilodrostat dose of 20 mg/day and oral supplementation was gradually reduced simultaneously with osilodrostat dose escalation. The reason why such severe hypokalemia occurred with low doses of osilodrostat and did not deteriorate further seems complex. One possible reason is the administration of high doses of potassium-saving antihypertensive drugs such as spironolactone and the angiotensin II receptor antagonist telmisartan. Additionally, one can consider other possible mechanisms, such as downregulation of the receptors of deoxycorticosterone (DOC) or other adrenal hormone precursors. However, this hypothesis requires further research and confirmation. Such an improvement of the potassium level during osilodrostat dose escalation was previously demonstrated in a patient with CD.11 Interestingly, in our Patient 2, no potassium supplementation was required during the whole time of osilodrostat therapy, although the doses were increased intensively up to the finally effective dose, which was the same (45 mg/day) as for Patient 1. In Patient 2, no actual response to doses lower than 20 mg/day was observed. UFC normalization was achieved after a week of administration of 45 mg/day, five weeks from the beginning of therapy. Although UFC normalization is not always required in pre-surgical treatment, clinical symptoms significantly improved in our patients only after the UFC upper normal level was achieved.
The present paper is one of only a few reports focused on osilodrostat therapy in CPA, and the only one presenting a different therapy course as compared to patients with CD. No case of CPA resistance to low doses of osilodrostat has been described. It should be underlined that in our report “low doses” of osilodrostat were higher than the average mean doses of osilodrostat used in clinical trials in patients with CD.3–6 Therefore, they should not generally be considered low but only much lower than those which were effective in our patients. Malik and Ben-Shlomo presented a case of CPA treated with osilodrostat, with an immediate decrease in cortisol level at 4 mg/day and adrenal insufficiency symptoms after dose escalation to 8 mg/day.15 Similar to our two cases, their patient was a middle-aged female with normal results of all other adrenal parameters, such as renin, angiotensin, or metanephrine levels. However, a CT scan was not performed (or presented), while magnetic resonance imaging revealed an indeterminate adrenal gland mass without a typical contrast phase/out-of-phase dropout for adenoma.15 Therefore, different morphology of cortisol-secreting adrenal tumor can potentially be considered a reason of the different response to treatment. Tanaka et al performed a multicenter study on the efficacy and safety of osilodrostat in Japanese patients with non-CD Cushing’s syndrome.16 Five patients with CPA were included in the study, and none of them required osilodrostat doses higher than 10 mg/day to achieve UFC normalization. However, most of the patients presented by Tanaka et al were previously treated with metyrapone,16 whereas both of our patients were treatment-naive. Previous metyrapone therapy may be considered as a potential reason of better response to osilodrostat. This hypothesis was confirmed in the quoted study by Tanaka et al, who demonstrated that at week 12 the median percent changes in the mUFC values were higher in patients previously treated with metyrapone (–98.97%) than in treatment-naive cases (–86.65%).16 Detomas et al performed a comparison of efficacy and safety of osilodrostat and metyrapone, with one CPA patients included in a group treated with osilodrostat, however no data on a dose required for a disease control are available separately for this particular patient.8 To the best of our knowledge, no more CPA cases have been described and therefore no further comparison is available.
Higher doses of osilodrostat were administered to a group of seven patients with hypercortisolism due to adrenocortical carcinoma (ACC) presented by Tabarin et al.17 A full control of hypercortisolism was achieved in one patient for each dose of 4, 8, 10, and 20 mg/day, and in three patients treated with 40 mg/day.17 These patients, however received other therapies including mitotane and chemotherapy, which can significantly modify the response to osilodrostat.
Several authors have reported the phenomenon of a partial or total loss of response to osilodrostat.5,16,17 In such cases, a response to treatment was initially achieved and then lost during treatment with the same dose. A further increase in osilodrostat dose usually resulted in the response resumption.5,16,17 Such a situation could not be suspected in either of our cases.
The presented cases provide a novel insight into modalities of treatment with osilodrostat in patients with CPA and demonstrate for the first time that an inverse cortisol response is possible in CPA cases, especially those with a higher CT density of adrenal adenoma. Such a situation should not be considered a contraindication to dose escalation. Conversely, the dose should be increased more intensively so as to achieve the initial efficacy threshold, which was 20 mg/day in both of our patients. The fully efficient dose that allowed UFC normalization was more than twice as high (45 mg/day in both cases). A similar approach should be applied in patients who do not respond to lower doses, such as Patient 2. The safety of osilodrostat therapy is strictly individual and not dose dependent in patients with CPA. Adverse events, including hypokalemia, severe hypertension, and edema, can be of life-threatening severity or may not occur regardless of the dose. Moreover, AEs of high severity may decrease with osilodrostat dose escalation. Our study demonstrated that osilodrostat is efficient and can be used in patients with CPA as a pre-surgical therapy if surgery is contraindicated due to hypercortisolism complications.
Our study presented two cases of CPA treated with osilodrostat, and a small size of our group is the main limitation of this report. Future research is required to confirm our observations.
Conclusion
In some patients with CPA, the doses of osilodrostat required for disease control can be much higher than those previously reported. Acceleration of the dose increase can be fast, and the risk of overdosing, adrenal insufficiency, and later necessity of dose reduction seem to be much lower than it could be expected. Low initial doses (<20 mg/day in our study) can be entirely ineffective or can even cause exacerbation of hypercortisolism, whereas high doses (45 mg/day in the present study) are efficient in pre-surgery UFC normalization. AEs associated with osilodrostat can be rapid, with severe hypokalemia despite active potassium supplementation, or may not occur even if high doses of osilodrostat are applied. Therefore, close monitoring for potential AEs is necessary.
Acknowledgments
The abstract included some parts of this paper was presented at the European Congress of Endocrinology ECE2023 as a rapid communication. The abstract was published in the Endocrine Abstracts Vol. 90 [https://www.endocrine-abstracts.org/ea/0090/].
Funding
The publication of this report was financially supported by the statutory funds of the Polish Mother’s Memorial Hospital – Research Institute, Lodz, Poland.
Disclosure
Professor Przemysław Witek reports personal fees from Investigator in the clinical trials paid by Novartis and Recordati Rare Diseases, outside the submitted work; lectures fees from Recordati Rare Diseases, Strongbridge, IPSEN. The authors report no other conflicts of interest in this work.
References
1. Fleseriu M, Auchus R, Bancos I, et al. Consensus on diagnosis and management of Cushing’s disease: a guideline update. Lancet Diabetes Endocrinol. 2021;9(12):847–875. doi:10.1016/S2213-8587(21)00235-7
2. Pivonello R, Isidori AM, De Martino MC, et al. Complications of Cushing’s syndrome: state of the art. Lancet Diabetes Endocrinol. 2016;4(7):611–629. doi:10.1016/S2213-8587(16)00086-3
3. Pivonello R, Fleseriu M, Newell-Price J, et al. Efficacy and safety of osilodrostat in patients with Cushing’s disease (LINC 3): a multicentre Phase III study with a double-blind, randomised withdrawal phase. Lancet Diabetes Endocrinol. 2020;8(9):48–761. doi:10.1016/S2213-8587(20)30240-0
4. Fleseriu M, Newell-Price J, Pivonello R, et al. Long-term outcomes of osilodrostat in Cushing’s disease: LINC 3 study extension. Eur J Endocrinol. 2022;187(4):531–541. doi:10.1530/EJE-22-0317
5. Fleseriu M, Biller BMK, Bertherat J, et al. Long-term efficacy and safety of osilodrostat in Cushing’s disease: final results from a Phase II study with an optional extension phase (LINC 2). Pituitary. 2022;25(6):959–970. doi:10.1007/s11102-022-01280-6
6. Gadelha M, Bex M, Feelders RA, et al. Randomized trial of osilodrostat for the treatment of Cushing disease. J Clin Endocrinol Metab. 2022;107(7):e2882–e2895. doi:10.1210/clinem/dgac178
7. Daniel E, Aylwin S, Mustafa O, et al. Effectiveness of metyrapone in treating cushing’s syndrome: a retrospective multicenter study in 195 patients. J Clin Endocrinol Metab. 2015;100(11):4146–4154. doi:10.1210/jc.2015-2616
8. Detomas M, Altieri B, Deutschbein T, et al. Metyrapone versus osilodrostat in the short-term therapy of endogenous cushing’s syndrome: results from a single center cohort study. Front Endocrinol. 2022;13:903545. doi:10.3389/fendo.2022.903545
9. U.S. food and drug administration home page. Available from: https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatment-adults-cushings-disease. Accessed March 22, 2023.
10. Agency for health technology assessment and tariff system home page. Available from: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwj6ypGbsfT9AhUMzYsKHTgAD2EQFnoECA8QAQ&url=https%3A%2F%2Fbipold.aotm.gov.pl%2Fassets%2Ffiles%2Fwykaz_tli%2FRAPORTY%2F2020_010.pdf&usg=AOvVaw3P2Q85gwi3JcxKkW3uxfOb. Accessed March 22, 2022.
11. Dzialach L, Sobolewska J, Respondek W, et al. Cushing’s syndrome: a combined treatment with etomidate and osilodrostat in severe life-threatening hypercortisolemia. Hormones. 2022;21(4):735–742. doi:10.1007/s42000-022-00397-4
12. Ekladios C, Khoury J, Mehr S, et al. Osilodrostat-induced adrenal insufficiency in a patient with Cushing’s disease. Clin Case Rep. 2022;10(11):e6607. doi:10.1002/ccr3.6607
13. Fleseriu M, Biller BMK. Treatment of Cushing’s syndrome with osilodrostat: practical applications of recent studies with case examples. Pituitary. 2022;25(6):795–809. doi:10.1007/s11102-022-01268-2
14. Summary of product characteristics. Available from: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwim1_KdsvT9AhVq-ioKHUZKAc4QFnoECA4QAQ&url=https%3A%2F%2Fwww.ema.europa.eu%2Fen%2Fdocuments%2Fproduct-information%2Fisturisa-epar-product-information_pl.pdf&usg=AOvVaw0S8nayCTdqNh1LsEcXVLEu. Accessed March 24, 2023.
15. Malik RB, Ben-Shlomo A. Adrenal cushing’s syndrome treated with preoperative osilodrostat and adrenalectomy. AACE Clin Case Rep. 2022;8(6):267–270. doi:10.1016/j.aace.2022.10.001
16. Tanaka T, Satoh F, Ujihara M, et al. A multicenter, Phase 2 study to evaluate the efficacy and safety of osilodrostat, a new 11β-hydroxylase inhibitor, in Japanese patients with endogenous Cushing’s syndrome other than Cushing’s disease. Endocr J. 2020;67(8):841–852. doi:10.1507/endocrj.EJ19-0617
17. Tabarin A, Haissaguerre M, Lassole H, et al. Efficacy and tolerance of osilodrostat in patients with Cushing’s syndrome due to adrenocortical carcinomas. Eur J Endocrinol. 2022;186(2):K1–K4. doi:10.1530/EJE-21-1008
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License. By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License. By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
-
 1
1
-
-
From all the news items I share, and the patients I talk to, it seems like not a lot of advancement is being made over the years. I hear about more people thinking that they have Cushing's after reading about some of the symptoms but it still seems hard for them to get to good doctors, to get diagnosed

Hopefully, next year will be better news!
-
 1
1
-
-
Even though I posted this in 2022 - the original website is still being redone. Probably it will be next year, too! LOL
-
 1
1
-
-
Abstract
Disclosure: C.M. Godar: None. E.B. Noble: None. N.O. Vietor: None. T.S. Knee: None.
Background: Cushing’s syndrome may rarely present as an emergency known as Florid Cushing’s Syndrome. Patients can exhibit severe hyperglycemia, hypertension, hypokalemia, infections, and hypercoagulability. Cushing’s syndrome is a rare disease, and the constellation of clinical features can be overlooked if clinicians are not aware of the manifestations of hypercortisolism. We present the case of a patient with Cushing’s syndrome that went unrecognized with life-threatening sequelae.
Case presentation: A 52-year-old woman with well-controlled type 2 diabetes and hypertension was admitted to the hospital for severe left lower extremity cellulitis. Prior to hospitalization she had noted rapid weight gain, fatigue, weakness, mental clouding, and moodiness. She was admitted for antibiotics and surgical debridement. The infection persisted despite broad spectrum antibiotics, multiple surgical debridements, and skin grafting. She became bacteremic, and extremity amputation was considered. She additionally developed hypertensive emergency, refractory hypokalemia, and hyperglycemia to 396 mg/dL. Exam was notable for facial plethora, supraclavicular fullness, dorsocervical fat pad, and violaceous abdominal striae. Cushing’s Syndrome was suspected, and labs revealed a significantly elevated random serum cortisol of 60.5mcg/dL (Ref 6.2-19.4), significantly elevated 24H urine cortisol of 2157mcg/24H (Ref 0-50), and ACTH elevated to 81.8pg/mL (Ref 7.2-63.3) that confirmed Cushing’s Disease. MRI sella and octreotide scans did not localize a lesion. Inpatient therapy included multiple antihypertensive agents, insulin drip, aggressive potassium repletion, and initiation of ketoconazole to reduce cortisol levels. Ketoconazole was maximally dosed and she underwent surgical exploration and removal of a small pituitary microadenoma. Following surgery, she developed transient adrenal insufficiency requiring hydrocortisone and she no longer required antihypertensives, insulin, or potassium therapy. Follow up 7 years later has revealed no recurrence of Cushing’s Disease.
Discussion: Cushing’s Syndrome may present with a variety of clinical features and rarely may present as a medical emergency. Delay in diagnosis can lead to Florid Cushing’s Syndrome which carries high risk for morbidity and mortality. This case illustrates the need for clinician awareness of the features of Cushing’s Syndrome: hypertension, hyperglycemia, rapid weight gain, cushingoid exam features, hypokalemia, hirsutism, virilization, infection, and/or hypercoagulable state. Severe hypercortisolism was responsible for this patient’s refractory infection, and if not controlled, she likely would have endured a lower extremity amputation. Rapid detection with elevated random serum and/or urine cortisol and treatment with a cortisol-lowering agent is critical and lifesaving.
Presentation: Thursday, June 15, 2023
This content is only available as a PDF.-
 1
1
-
-
Two years later
“My Dream Day“…
I’d wake up refreshed and really awake at about 7:00AM and
take the dog out(no more dog )
) for a brisk run. Maybe do a Silver Sneakers online workout unless it's a water aerobics day.Get home about 8:00AM and start on my website work.
Later in the morning, I’d get some bills paid – and there would be enough money to do so!
After lunch,
out with the dog again, then practice the piano some, practice my balalaika and tenor recorder son, read a bit, finish up the website work, teach a few piano students, work on my church job, then dinner.After dinner, check email,
out with the dog, maybe handbell or choir practice, a bit of TV, then bed about 10PMNothing fancy but NO NAPS. Work would be getting done, time for hobbies,
the dog, 3 healthy meals.Just a normal life that so many take for granted. Or, do they?
I would love to have enough money to retire from some of those jobs, to travel more, to cruise more...
-
Highlights
- Aim to identify independent risk factors for postoperative delirium after pituitary adenoma surgery.
- Select matched subjects by Propensity Score Matching to reduce potential biases caused by variables.
- Enhance preoperative communication to minimize the occurrence of delirium, for patients at high risk of postoperative delirium.
- Minimize surgery duration and general anesthesia, optimize perioperative sedation regimen.
- Reducing unnecessary or excessive protective physical restraints.
Abstract
Objectives
The primary aim of this study is to explore the factors associated with delirium incidence in postoperative patients who have undergone endoscopic transsphenoidal approach surgery for pituitary adenoma.
Methods
The study population included patients admitted to Tianjin Huanhu Hospital's Skull Base Endoscopy Center from January to December 2022, selected through a retrospective cohort study design. The presence of perioperative delirium was evaluated using the 4 'A's Test (4AT) scale, and the final diagnosis of delirium was determined by clinicians. Statistical analysis included Propensity Score Matching (PSM), χ2 Test, and Binary Logistic Regression.
Results
A total of 213 patients were included in this study, and the incidence of delirium was found to be 29.58 % (63/213). Among them, 126 patients were selected using PSM (delirium:non-delirium = 1:1), ensuring age, gender, and pathology were matched. According to the results of univariate analysis conducted on multiple variables, The binary logistic regression indicated that a history of alcoholism (OR = 6.89, [1.60–29.68], P = 0.010), preoperative optic nerve compression symptoms (OR = 4.30, [1.46–12.65], P = 0.008), operation time ≥3 h (OR = 5.50, [2.01–15.06], P = 0.001), benzodiazepines for sedation (OR = 3.94, [1.40–11.13], P = 0.010), sleep disorder (OR = 3.86, [1.40–10.66], P = 0.009), and physical restraint (OR = 4.53, [1.64–12.53], P = 0.004) as independent risk factors for postoperative delirium following pituitary adenoma surgery.
Conclusions
For pituitary adenoma patients with a history of alcoholism and presenting symptoms of optic nerve compression, as well as an operation time ≥3 h, enhancing communication between healthcare providers and patients, improving perioperative sleep quality, and reducing physical restraint may help decrease the incidence of postoperative delirium.
Introduction
In clinical practice, patients admitted to the intensive care unit (ICU) during the postoperative period after endoscopic transsphenoidal tumorectomy of pituitary adenoma often experience episodes of delirium. According to a recent retrospective analysis conducted at a single center, the incidence of postoperative delirium among these patients was found to be 10.34 % (n = 360) [1]. Delirium is a common complication following neurosurgery, characterized by acute distraction, confusion in thinking, sleep disorders, and cognitive decline. The incidence of delirium in admitted patients after neurosurgery has been reported to be 19 %, with a range of 12 % to 26 % depending on clinical features and the methods used for delirium assessment [2], [3], [4]. The incidence of postoperative delirium varied across different types of neurosurgical diseases, as reported in a meta-analysis [2]. Specifically, the incidences were 8.0 % for patients with neurological tumors, 20 % for those undergoing functional neurosurgery, 24.0 % for microvascular decompression patients, 19.0 % for traumatic brain injury patients, 42.0 % for neurovascular patients, and 17.0 % for the mixed population undergoing neurosurgery procedures. Furthermore, the incidence rates of delirium in intensive care units (ICUs), general wards, or both combined were found to be 24.0 %, 17 %, and 18 %, respectively.
The aforementioned issue not only leads to prolonged hospital stays and increased healthcare costs, but also exerts a significant impact on patient consciousness and cognitive function. Therefore, early and accurate identification of delirium in post-neurosurgical patients is crucial. However, due to frequent co-occurrence with primary brain injury, related complications can also lead to cognitive impairment or even decreased levels of consciousness, posing challenges for timely and precise identification of delirium. Currently, the primary focus lies in the prevention of delirium within the neurosurgical ICU setting. Early identification and comprehensive pre-surgical assessment are positively significant measures for preventing postoperative delirium occurrence [5], [6]. In this study, a retrospective cohort design was employed to collect pertinent data and statistically analyze the incidence of delirium, as well as its associated influencing factors, among patients admitted to the neurosurgical ICU for pituitary adenoma treatment. And now it is reported as follows.
Section snippets
Patient selection
A retrospective cohort study design was employed to select 213 pituitary adenomas admitted to the Skull Base and Endoscopy Center of Tianjin Huanhu Hospital between January 2022 and December 2022 as the subjects for investigation, with a review of their medical records. The mean age was (50.03 ± 15.72) years, ranging from 20–79 years old (Fig. 1). Informed consent was obtained from all patients or their families, ensuring compliance with the requirements stated in the Declaration of Helsinki.
Inclusion criteria
a.
Propensity score matching
The present study enrolled a total of 213 patients with pituitary tumors, among whom 63 exhibited symptoms related to delirium while the remaining 150 did not. Consequently, the incidence rate of delirium was determined to be 29.58 % in this cohort of patients admitted to the intensive care unit following pituitary tumor surgery. The univariate analysis revealed no significant differences in age (≥65y old, 23.8 % vs. 23.3 %, P = 0.940) and gender (male, 49.2 % vs. 56.7 %, P = 0.318) between the
Background of perioperative delirium in transsphenoidal endoscopic pituitary adenoma surgery
The pituitary gland is situated within the sella turcica and comprises two distinct components. The anterior pituitary, known as the adenohypophysis, functions as an endocrine organ responsible for secreting growth hormone, prolactin, adrenocorticotropic hormone, thyrotropin, follicle-stimulating hormone and luteinizing hormone. On the other hand, the posterior pituitary, referred to as the neurohypophysis, serves as a direct extension of the hypothalamus and acts as a storage site for
Conclusions
To enhance the evaluation of postoperative patients at risk of delirium, it is anticipated that optimizing doctor-nurse-patient communication and minimizing unnecessary and indiscriminate protective measures will mitigate the incidence of delirium following pituitary tumor surgery. This study is a single-center prospective study conducted at our institution, which has several inherent limitations. A large-scale multicenter prospective study is anticipated to further investigate the associated
Limitations
There are multiple factors that influence the occurrence of delirium following neurosurgery. This retrospective study solely focused on analyzing and comparing general patient data, medical history, and potential perioperative factors contributing to delirium, without considering any other known or unknown variables in this analysis. The pituitary gland functions as a neuroendocrine organ involved in the regulation of neuroendocrine processes. Changes in hormone levels following surgery for
Funding
All authors affirm that this study was conducted without any fund support from external organizations.
CRediT authorship contribution statement
Shusheng Zhang: Writing – original draft, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Yanan Chen: Writing – original draft, Investigation, Data curation. Xiudong Wang: Validation, Supervision, Project administration, Methodology, Conceptualization. Jun Liu: Software, Formal analysis, Data curation. Yueda Chen: Validation, Supervision, Methodology, Investigation. Guobin Zhang: Writing – review & editing, Validation, Supervision, Methodology, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References (21)
-
G. Sousa et al.
Postoperative delirium in patients with history of alcohol abuse
Rev Esp Anestesiol Reanim
(2017) -
M. Terzaghi et al.
Sleep disorders and acute nocturnal delirium in the elderly: a comorbidity not to be overlooked
Eur J Intern Med
(2014) -
S. Lee et al.
Opioid and benzodiazepine use in the emergency department and the recognition of delirium within the first 24 hours of hospitalization
J Psychosom Res
(2022) -
A.J. Slooter et al.
Delirium in critically ill patients
Handb Clin Neurol
(2017) -
E. Wang et al.
Effect of perioperative benzodiazepine use on intraoperative awareness and postoperative delirium: a systematic review and meta-analysis of randomized controlled trials and observational studies
Br J Anaesth
(2023) -
E. Rollo et al.
Physical restraint precipitates delirium in stroke patients
J Neurol Sci
(2021) -
H. Chen et al.
The incidence and predictors of postoperative delirium after brain tumor resection in adults: A cross-sectional survey
World Neurosurg
(2020) -
J. Liu et al.
Identifying hormones and other perioperative risk factors for postoperative delirium after endoscope-assisted transsphenoidal pituitary adenoma resection: A retrospective, matched cohort study
Brain Behav
(2023) -
P.R. Kappen et al.
Delirium in neurosurgery: a systematic review and meta-analysis
Neurosurg Rev
(2022) -
J. Wang et al.
Risk factors for the incidence of delirium in cerebrovascular patients in a Neurosurgery Intensive Care Unit: A prospective study
J Clin Nurs
(2018)
There are more references available in the full text version of this article. -
Happy Birthday, Harvey!
-
 1
1
-
-
It's Sunday again, so this is another semi-religious post so feel free to skip it

I'm sure that many would think that Abide With Me is a pretty strange choice for my all-time favorite hymn, especially since it often shows up at funerals and memorial services.
My dad was a Congregational (now United Church of Christ) minister so I was pretty regular in church attendance in my younger years.
Some Sunday evenings, he would preach on a circuit and I'd go with him to some of these tiny churches. The people there, mostly older folks, liked the old hymns best - Fanny Crosby and so on.
So, some of my "favorite hymns" are those that I sang when I was out with my Dad. Fond memories from long ago.
In 1986 I was finally diagnosed with Cushing's after struggling with doctors and trying to get them to test for about 5 years. I was going to go into the NIH (National Institutes of Health) in Bethesda, MD for final testing and then-experimental pituitary surgery.
I was terrified and sure that I wouldn't survive the surgery.
Somehow, I found a 3-cassette tape set of Reader's Digest Hymns and Songs of Inspiration and ordered that. The set came just before I went to NIH and I had it with me.
At NIH I set up a daily "routine" of sorts and listening to these tapes was a very important part of my day and helped me get through the ordeal of more testing, surgery, post-op and more.
When I had my kidney cancer surgery, those tapes were long broken and irreplaceable, but I had replaced all the songs - this time on my iPod.
Abide With Me was on this original tape set and it remains a favorite to this day. Whenever we have an opportunity in church to pick a favorite, my hand always shoots up and I request page 700. When someone in one of my handbell groups moves away, we always sign a hymnbook and give it to them. I sign page 700.
I think that many people would probably think that this hymn is depressing. Maybe it is but to me it signifies times in my life when I thought I might die and I was so comforted by the sentiments here.
This hymn is often associated with funeral services and has given hope and comfort to so many over the years - me included.
If you abide in Me, and My words abide in you, you will ask what you desire, and it shall be done for you.
~John 15:7
Abide With Me
Words: Henry F. Lyte, 1847.
Music: Eventide, William H. Monk, 1861. Mrs. Monk described the setting:
This tune was written at a time of great sorrow—when together we watched, as we did daily, the glories of the setting sun. As the last golden ray faded, he took some paper and penciled that tune which has gone all over the earth.
Lyte was inspired to write this hymn as he was dying of tuberculosis; he finished it the Sunday he gave his farewell sermon in the parish he served so many years. The next day, he left for Italy to regain his health. He didn’t make it, though—he died in Nice, France, three weeks after writing these words. Here is an excerpt from his farewell sermon:
O brethren, I stand here among you today, as alive from the dead, if I may hope to impress it upon you, and induce you to prepare for that solemn hour which must come to all, by a timely acquaintance with the death of Christ.
For over a century, the bells of his church at All Saints in Lower Brixham, Devonshire, have rung out “Abide with Me” daily. The hymn was sung at the wedding of King George VI, at the wedding of his daughter, the future Queen Elizabeth II, and at the funeral of Nobel peace prize winner Mother Teresa of Calcutta in1997.
Abide with me; fast falls the eventide;
The darkness deepens; Lord with me abide.
When other helpers fail and comforts flee,
Help of the helpless, O abide with me.
Swift to its close ebbs out life’s little day;
Earth’s joys grow dim; its glories pass away;
Change and decay in all around I see;
O Thou who changest not, abide with me.
Not a brief glance I beg, a passing word;
But as Thou dwell’st with Thy disciples, Lord,
Familiar, condescending, patient, free.
Come not to sojourn, but abide with me.
Come not in terrors, as the King of kings,
But kind and good, with healing in Thy wings,
Tears for all woes, a heart for every plea—
Come, Friend of sinners, and thus bide with me.
Thou on my head in early youth didst smile;
And, though rebellious and perverse meanwhile,
Thou hast not left me, oft as I left Thee,
On to the close, O Lord, abide with me.
I need Thy presence every passing hour.
What but Thy grace can foil the tempter’s power?
Who, like Thyself, my guide and stay can be?
Through cloud and sunshine, Lord, abide with me.
I fear no foe, with Thee at hand to bless;
Ills have no weight, and tears no bitterness.
Where is death’s sting? Where, grave, thy victory?
I triumph still, if Thou abide with me.
Hold Thou Thy cross before my closing eyes;
Shine through the gloom and point me to the skies.
Heaven’s morning breaks, and earth’s vain shadows flee;
In life, in death, O Lord, abide with me.
http://cushieblog.files.wordpress.com/2012/04/maryo-butterfly-script1.gif
-
 1
1
-




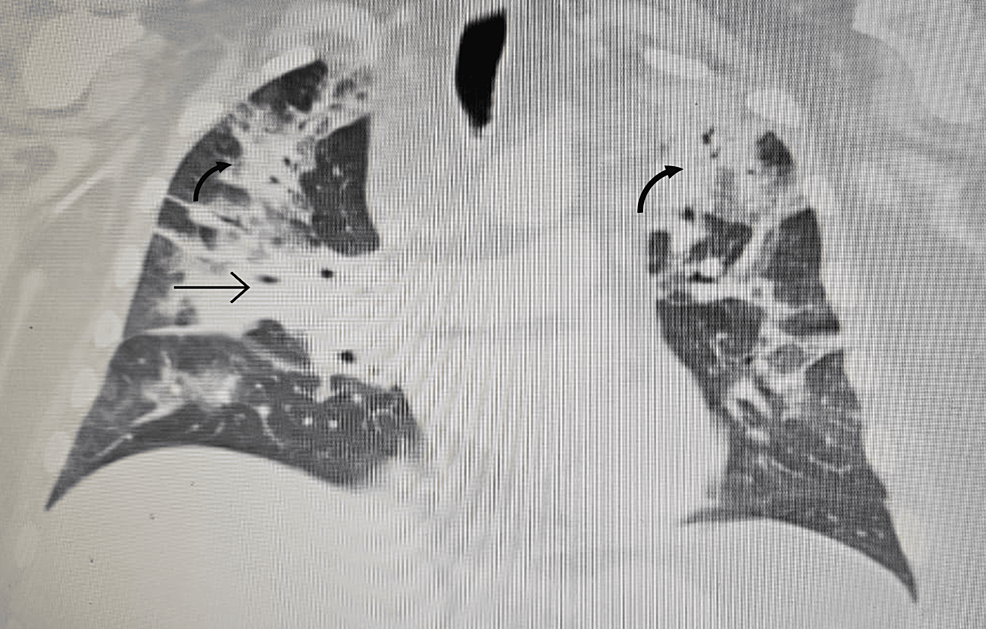
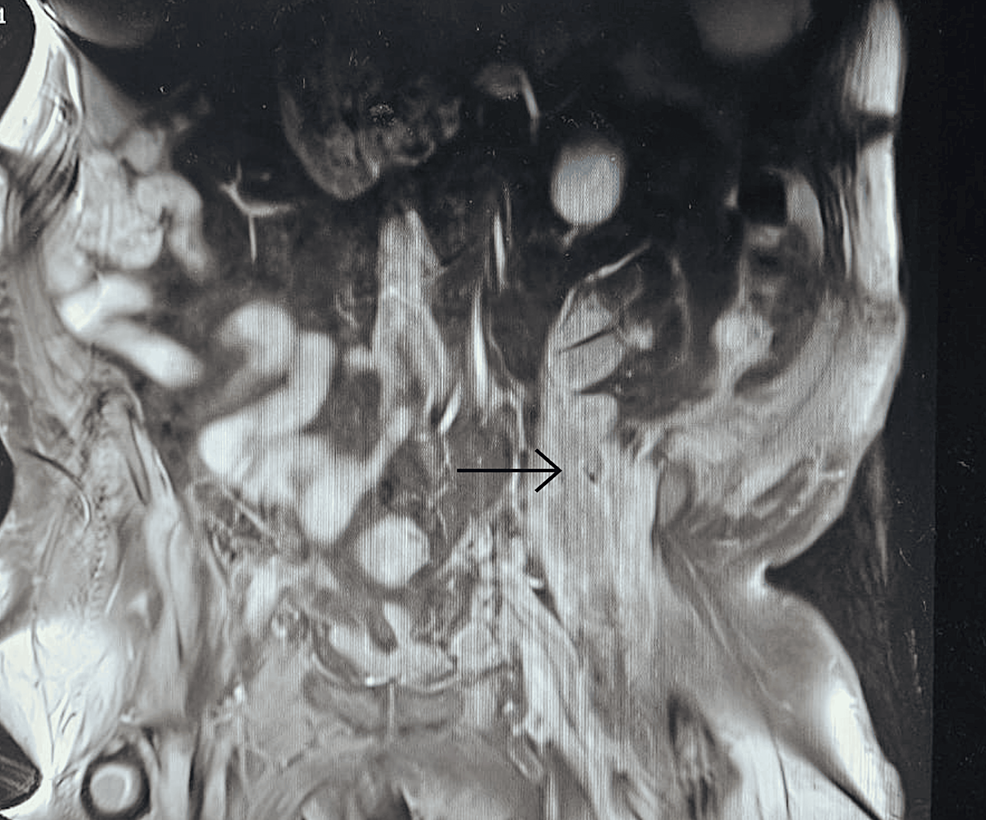
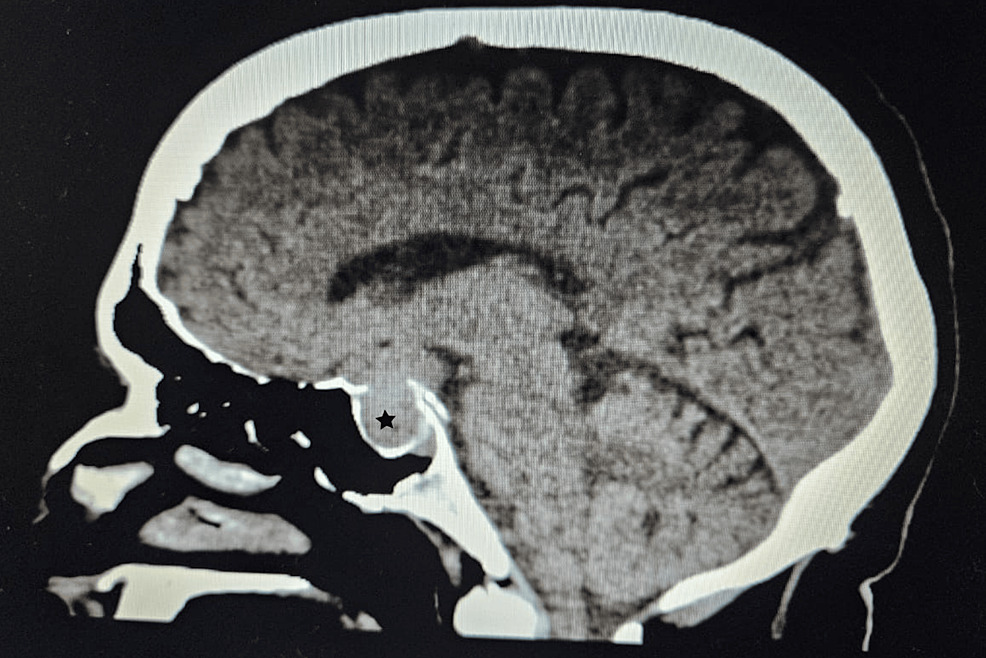




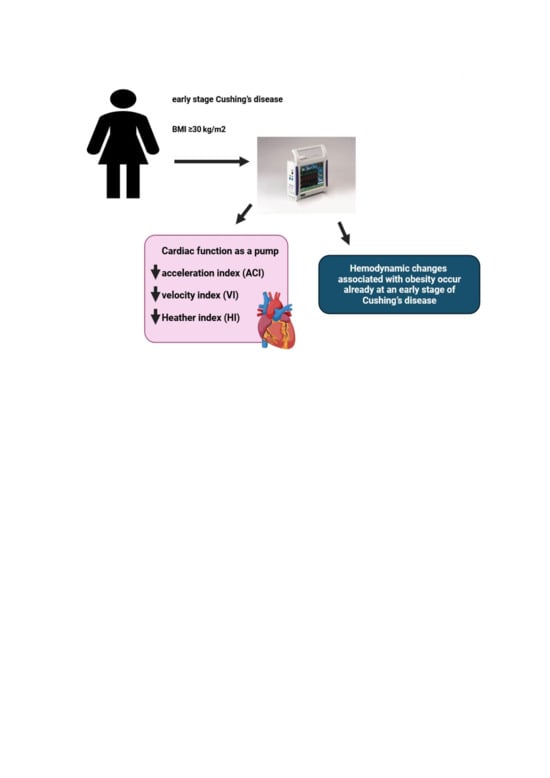

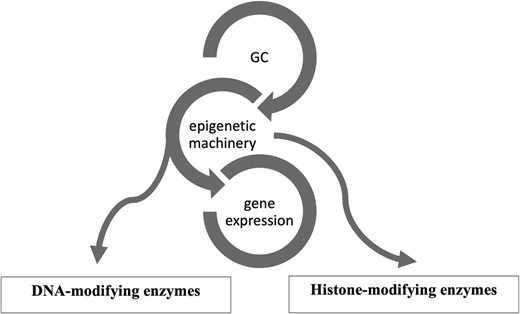
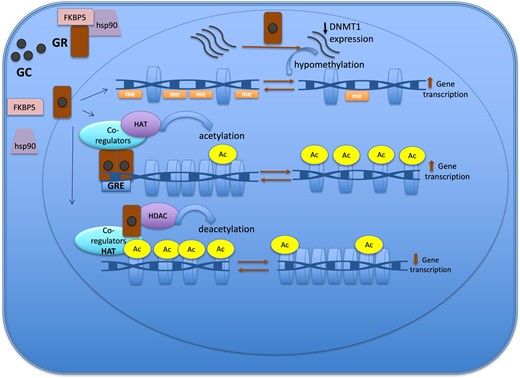

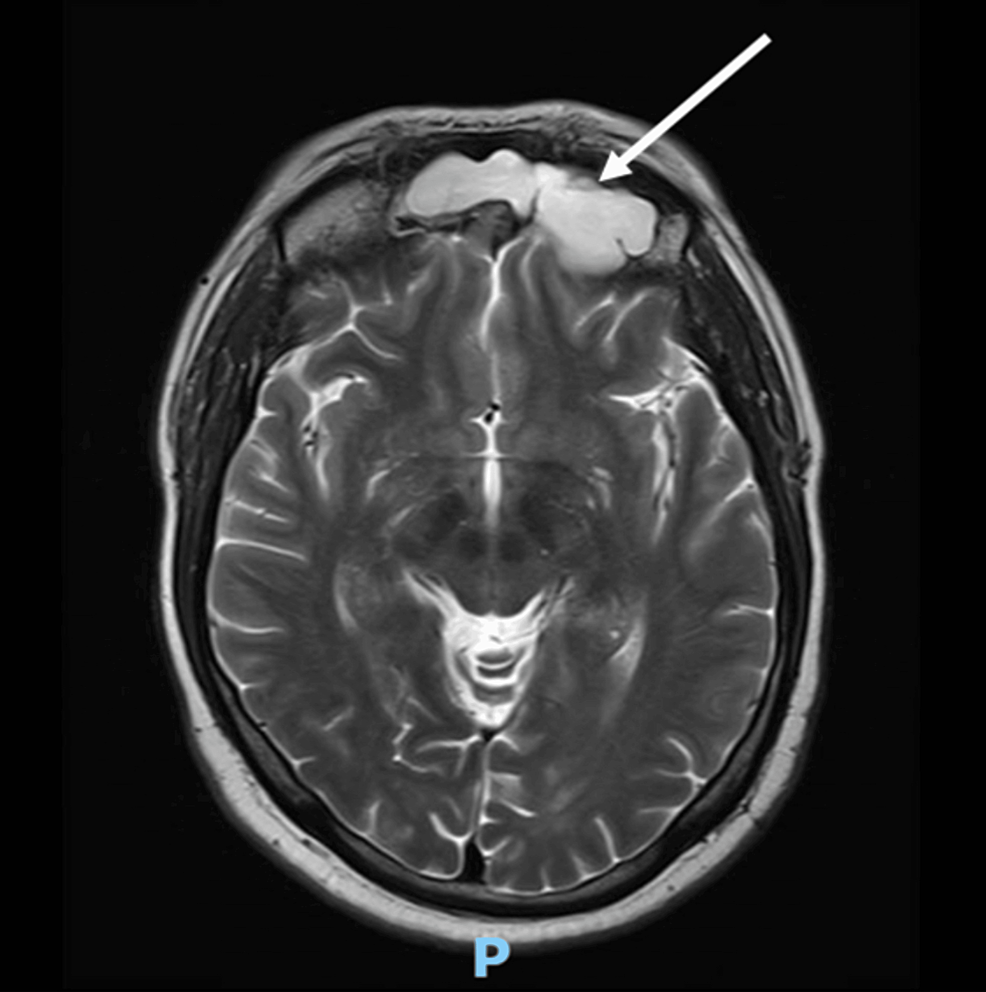
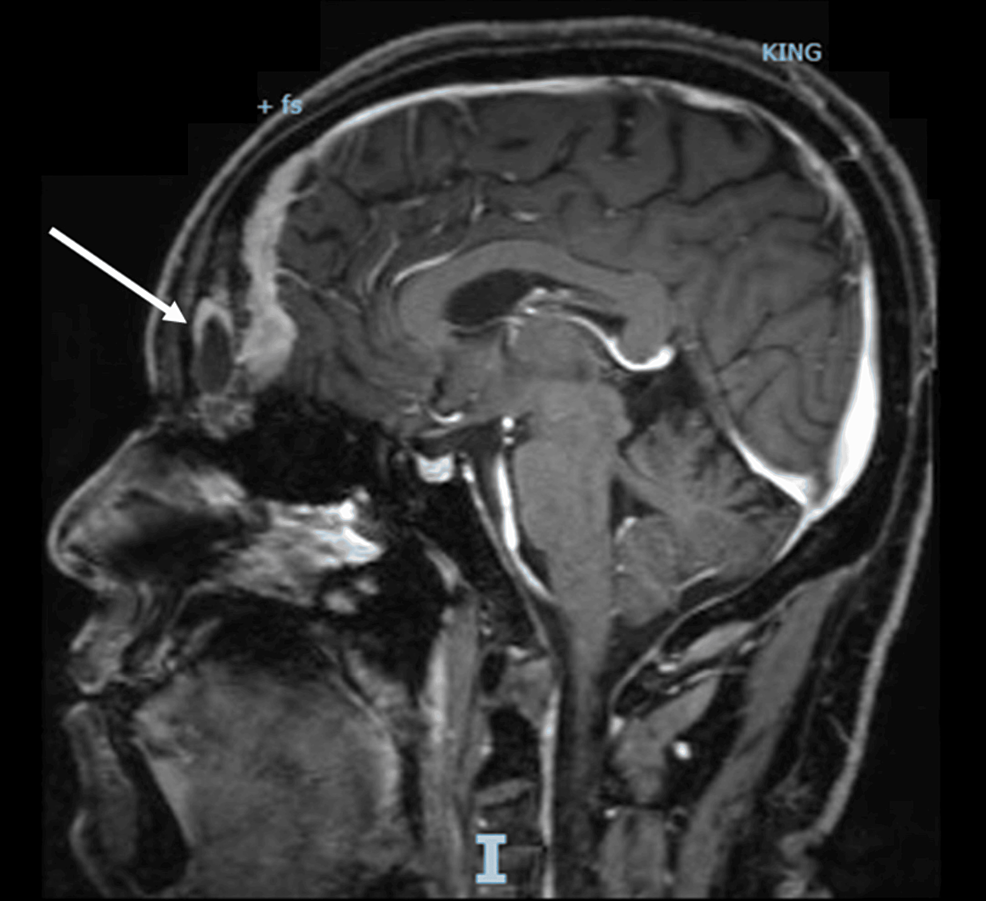
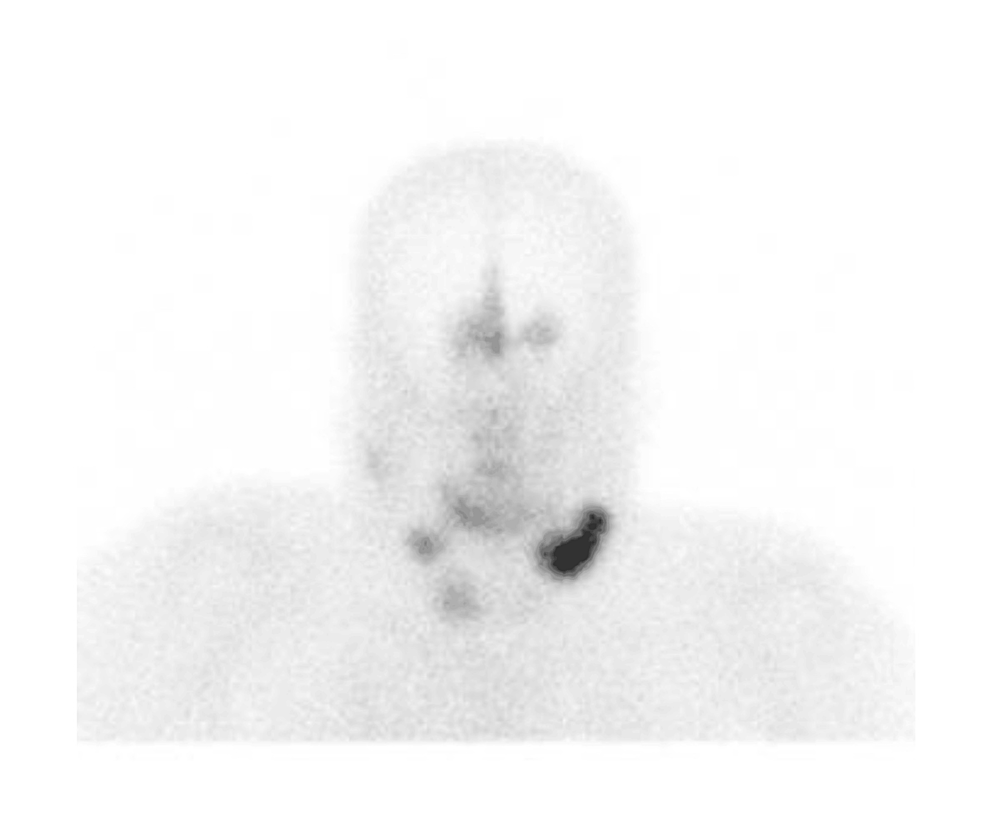
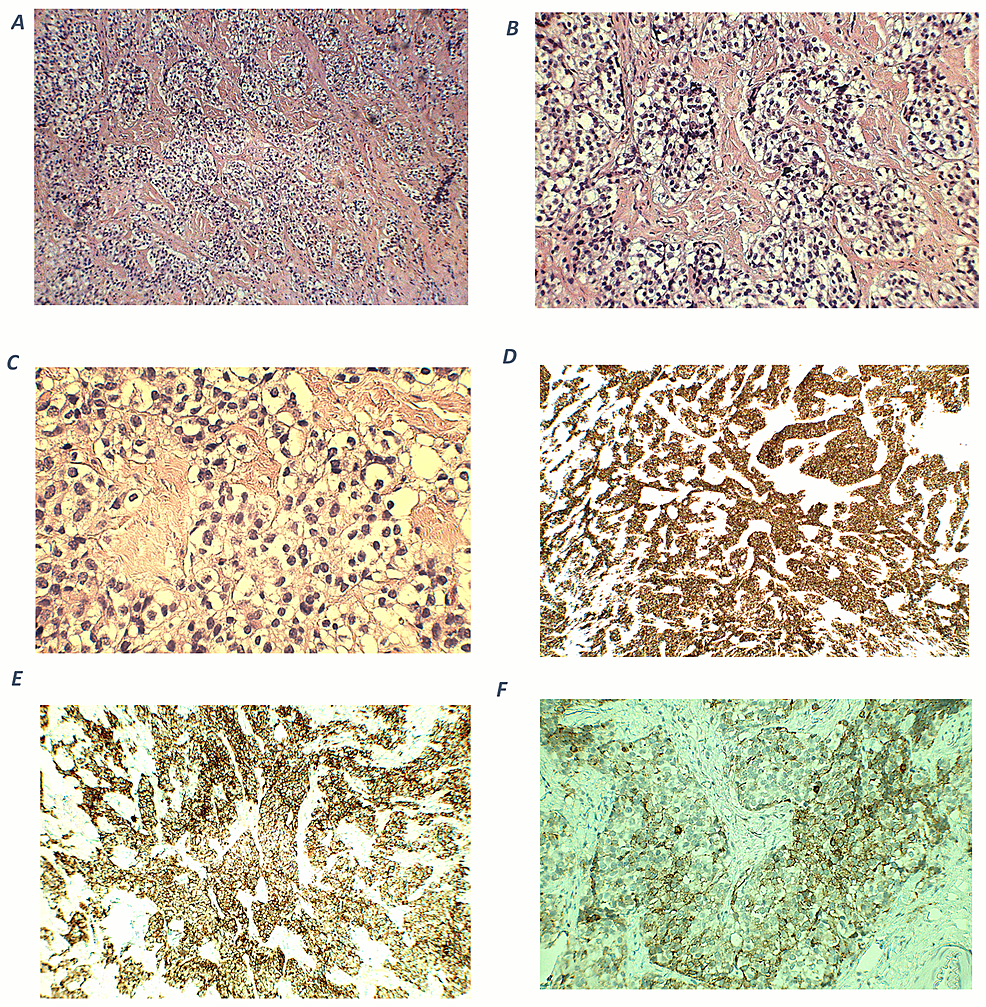


Day 24: Cushing’s Awareness Challenge
in Cushing's Awareness Day, April 8
Posted
Over the years, we went on several Windjammer Barefoot Cruises. We liked them because they were small, casual and were fairly easy on the wallet.
They sailed around the Caribbean to a variety of islands, although they sometimes changed itineraries depending on weather, crew, whatever. One trip we were supposed to go to Saba but couldn't make port. A lot of people got off at the next port and flew home.
The captains were prone to "Bedtime Stories" which were often more fiction than true but they added to the appeal of the trip. We didn't care if we missed islands or not - we were just there to sail over the waves and enjoy the ride.
The last trip we took with them was about two years before I started having Cushing's problems. (You wondered how I was going to tie this together, right?)
The cruise was uneventful, other than the usual mishaps like hitting docks, missing islands and so on. Until it was a particularly rough sea one day. I was walking somewhere on deck and suddenly a wave came up over the deck making it very slippery. I fell and cracked the back of my head on the curved edge of a table in the dining area. I had the next-to-the-worse headache I have ever had, the worst being after my pituitary surgery. At least after the surgery, I got some morphine.
We asked several doctors later if that hit could have contributed to my Cushing's but doctors didn't want to get involved in that at all.
The Windjammer folks didn't fare much better, either. In October 1998, Hurricane Mitch was responsible for the loss of the s/v Fantome (the last one we were on). All 31 crew members aboard perished; passengers and other crew members had earlier been offloaded in Belize.
The story was recorded in the book The Ship and the Storm: Hurricane Mitch and the Loss of the Fantome by Jim Carrier. The ship, which was sailing in the center of the hurricane, experienced up to 50-foot (15 m) waves and over 100 mph (160 km/h) winds, causing the Fantome to founder off the coast of Honduras.
"In October 1998, the majestic schooner Fantome came face-to-face with one of the most savage storms in Atlantic history. The last days of the Fantome are reconstructed in vivid and heartbreaking detail through Jim Carrier's extensive research and hundreds of personal interviews. What emerges is a story of courage, hubris, the agony of command, the weight of lives versus wealth, and the advances of science versus the terrible power and unpredictability of nature."
This event was similar to the Perfect Storm in that the weather people were more interested in watching the hurricane change directions than they were in people who were dealing with its effects.
I read this book and I was really moved by the plight of those crew members.
I'll never know if that hit on my head contributed to my Cushing's but I have seen several people mention on the message boards that they had a traumatic head injury of some type in their earlier lives.